Final results from the MARIPOSA trial provide further supporting evidence to the first-line treatment combination
New topline data from the MARIPOSA study suggests that amivantamab, a human bispecific antibody targeting epidermal growth factor receptor (EGFR), plus lazertinib, a brain-penetrant irreversible third-generation EGFR tyrosine kinase inhibitor (TKI), is expected to extend life by a median of one year or more in patients with untreated epidermal growth factor receptor (EGFR)-mutated non-small cell lung cancer (NSCLC) compared to the treatment with osimertinib (Abstract 4O). Overall survival (OS) results were presented at the European Lung Cancer Congress 2025 (ELCC) in March.
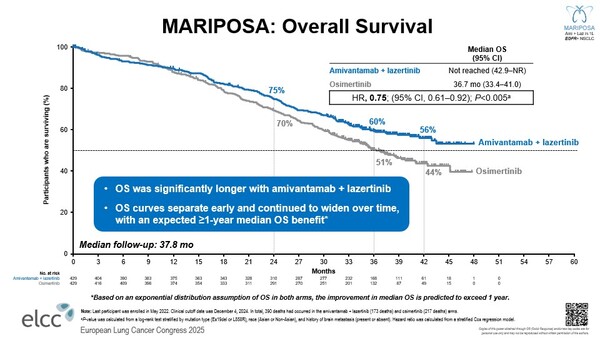
At a median follow up of 37.8 months, amivantamab plus lazertinib led to a statistically significant and clinically meaningful reduction in mortality compared to osimertinib (hazard ratio (HR) for death, 0.75; 95% CI, 0.61–0.92; P<0.005) (Figure). Based on an exponential distribution assumption of OS in both arms, the improvement in median OS with the combination therapy is predicted to exceed one year. At three years, 60% of participants were alive in the amivantamab plus lazertinib arm compared to 51% for osimertinib, and benefit continued at 42-months with survival rates of 56% and 44%, respectively.
Amivantamab plus lazertinib demonstrated a clinically meaningful improvement in intracranial prolonged-free survival (icPFS) and intracranianl duration of response (icDoR) compared to osimertinib (36% vs 18% and 35.7 vs 29.6 months, respectively).
Median duration of treatment was 27.0 months for the combination therapy and 22.4 months for osimertinib. Safety profile was consistent with the primary analysis. Adverse events (AEs), mostly EGFR- and MET-related and grades 1–2, occurred early and showed to be reduced with simple prophylactic approaches.
Discussing the data presented at ELCC 2025, Dr Maurice Pérol, Léon Bérard Cancer Center in Lyon, France, commented: “The amivantamab-lazertinib combination achieved a significant and a clinically relevant OS over osimertinib with an absolute 3-year benefit of 9%, demonstrating both the importance of frontline treatment and the lower impact of subsequent lines on survival and that preventing or delaying the emergence of acquired resistance is more effective than trying to overcome them. This came at the cost of significant, early and long-lasting toxicity, requiring aggressive management and impacting daily quality of life of patients during a period when the disease is generally well-controlled.”
“We have now two intensified first-line treatment options for patients with EGFR mutant advanced NSCLC, with the amivantamab-lazertinib combination and the FLAURA-2 regimen, i.e. osimertinib and chemotherapy”, Pérol concluded. “As with the FLAURA2 regimen, the benefit/risk ration of the amivantamab-lazertinib combination must take into account the treatment objectives shared with the patient, justified – but this is a personal opinion – to reserve the combination for the clinical and genomic profiles of high-risk disease and ideally with the support of additional biomarkers.”
Amivantamab in combination with lazertinib was approved by the U.S. Food and Drug Administration in 2024 and the European Medicines Agency in early 2025 for the first-line treatment of locally advanced or metastatic NSCLC with EGFR exon 19 deletions or exon 21 L858R substitution mutations. Decisions were based on the positive findings of the MARIPOSA trial on treatment-naïve patients receiving amivantamab–lazertinib, osimertinib, or lazertinib, which were firstly presented at the ESMO Congress 2023. The combination therapy demonstrated to reduce the risk of disease progression or death by 30% compared to osimertinib monotherapy (hazard ratio [HR] 0.70; 95% confidence interval [CI] 0.58–0.85; p<0.001), with a median PFS of 23.7 months (95% CI 19.1–27.7) and 16.6 months (95% CI 14.8–18.5), respectively (N Engl J Med. 2024 Jun 26). Improved durability of response among confirmed responders was also seen for amivantamab-lazertinib versus osimertinib (25.8 versus 16.8 months). Additionally, in patients with a history of brain metastases, amivantamab-lazertinib prolonged PFS versus osimertinib (18.3 versus 13.0 months; HR 0.69; 95% CI 0.53-0.92).