An improvement was observed when immunotherapy was added to neoadjuvant chemotherapy, but further data are needed to confirm the survival benefit
Final analysis data from the multicenter, single-arm phase II SAKK 16/14 trial showed that perioperative treatment with durvalumab in addition to neoadjuvant chemotherapy with cisplatin and docetaxel, followed by surgery led to an improvement of event-free survival (EFS) in 68 patients with resectable stage IIIA (N2) non-small cell lung cancer (NSCLC) compared to chemotherapy alone at a median follow up of 72 months. Results were reported at the European Lung Cancer Congress (ELCC) 2025 (Paris, 26–29 March) (Abstract 189MO).
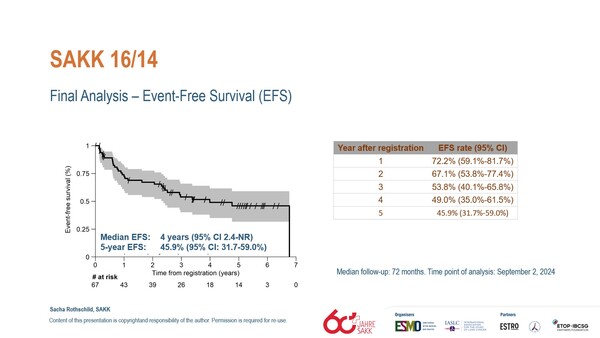
At data cut-off, the median EFS was 4.0 years (95% confidence interval (CI): 2.4 years-NR) and 5-year EFS rate was 45.9% (95% CI: 31.7–59.0%) (Figure). The median overall survival (OS) was not reached and estimated 5-year OS rate was 65.8% (95% CI: 52.9–76.0%). The addition of perioperative immunotherapy to the standard of care also resulted in a high major pathological response rate (MPR; <10% viable tumor cells) which was observed in 34 patients (62%), and 10 patients (18%) achieved a pathologic complete response (pCR). Postoperative nodal downstaging (ypN0-1) was reported in 37 patients (67%). Study authors announced that exploratory analyses of tissue and blood biomarkers are ongoing and a currently recruiting trial - SAKK 16/18 (NCT04245514) - is further exploring the potential benefit of immune-modulatory radiotherapy targeting the primary tumor.
Commenting on the results at ELCC 2025, Prof. Yolande Lievens, Ghent University Hospital, Belgium, noted that immune checkpoint inhibition in resectable stage III NSCLC is an evolving field and other three important studies investigating perioperative immunotherapy in this setting such as the Checkmate 77T (N Engl J Med. 2024 May 16;390(19):1756-1769), Keynote 671 (N Engl J Med. 2023 Aug 10;389(6):491-503) and AEGEAN (N Engl J Med. 2023 Nov 2;389(18):1672-1684) were recently published. Comparing the SAKK 16/14 trial to them, she added: “These three big trials have used a similar approach to induction giving chemo-immunotherapy for four cycles prior surgery, while the SAKK 16/14 study used chemo-immune checkpoint inhibition in a sequential way. They all aimed to give immune checkpoint inhibitors (ICIs) for one year after surgery. The SAKK 16/14 study had the longest follow up, but was also a small study in comparison to the other ones, it is important that it focusses on a very detailed group of patients – all IIIA (N2) - and this also give the opportunity to really analyse the impact on the pathological nodal downstaging which was in more than 67%. The pathological complete remission was comparable to the other randomised trials, while OS and EFS are difficult to compare because were not mature enough in the other studies.”
Lievens concluded that despite the results from the SAKK 16/14 trial are encouraging, they cannot influence clinical practice at the moment and that further research will need to answer some pending questions including what factors are behind Grade 3 toxicity of the regimen and the additional benefits in terms of quality of life and disease burden for patients.