Data reassures on targeting KRAS G12C mutations in older patients who reflect a large part of the real-world population seen in clinics
“Nowadays, mutations like KRAS G12C – the most common KRAS mutation in non-small cell lung cancer (NSCLC) – are routinely targeted in the clinic by small-molecule inhibitors. Novel effective agents and strategies are needed in this setting, but crucial to their development is ensuring that they are trialled in studies enrolling patients seen in real-world clinical settings,” explains Dr Silvia Novello of the University of Torino, Italy, commenting on subgroup data presented at the European Lung Cancer Congress 2026 (Copenhagen, 25–28 March).
The elderly and those with multiple comorbidities form a large contingent of patients with KRAS G12C-mutated NSCLC (J Clin Med. 2022;11:4098) but they are often underrepresented in trials. The elderly cancer patient has a unique set of needs including declining physical functionality, increased comorbidities, compromised hepatic and renal functions, pulmonary issues and haematopoietic disorders, as well as concerns related to surgical trauma and the adverse effects of chemoradiotherapy, along with economic and social considerations.
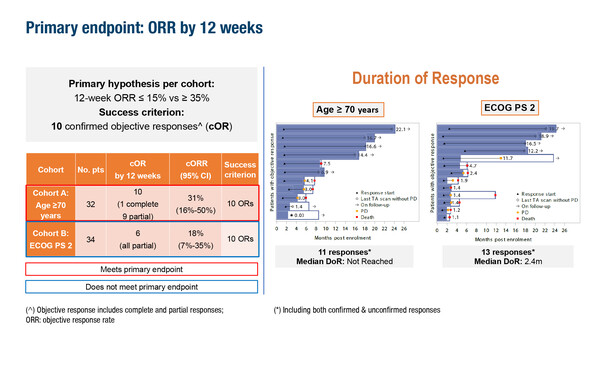
In final results from the phase II ETOP ADEPPT study investigating adagrasib in KRAS G12C-mutated NSCLC, presented in Copenhagen, the primary endpoint (≥10 patients achieving a confirmed objective response at 12 weeks) was achieved among patients aged ≥70 years with an ECOG performance status (PS) 0–1 (10/32; objective response rate [ORR] 31%) but not among patients aged ≥18 years with an ECOG PS 2 (6/34; ORR 18%) (Abstract 5MO, key results in the box below). Respective median progression-free survival (PFS) and overall survival were 7.6 months and 9.5 months for the older cohort, and 2.7 months and 4.3 months for the ECOG PS 2 cohort. At 12 weeks, quality of life (measured using the NCCN-Functional Assessment of Cancer Therapy Lung Symptom Index-17) had increased from baseline by 2.3 points in the older cohort and by 6.6 points in the ECOG PS 2 cohort.
“The results support the available literature and reassure clinicians that adagrasib can be beneficial in older patients with KRAS G12C-mutated NSCLC, but that caution is required in patients of any age with a poor performance status,” notes Novello. “The evident difference in outcomes observed in the study emphasises that the two cohorts, which are often evaluated under one umbrella in clinical research, actually demand individual investigation. However, the small numbers included in the trial speak to the difficulty of recruiting sufficient suitable patients and innovative ways must be sought to achieve this.”
Also presented in Copenhagen, updated results from the phase I KANDELIT-001 trial revealed that a combination of the next-generation, oral selective KRAS G12C-guanosine diphosphate (GDP) covalent inhibitor, MK-1084, with pembrolizumab led to encouraging activity (Abstract 4MO, key results in the box below). The ORR was 87% among 54 patients with previously untreated NSCLC and a PD-L1 tumour proportion score (TPS) ≥50% and 55% among 44 patients with a PD-L1 TPS 1–49%. Median progression-free survival (PFS) across all 98 patients was 28.9 months. Less-marked benefits were seen with MK-1084 plus pembrolizumab and chemotherapy in 46 previously untreated patients with NSCLC and any TPS (ORR 65%; PFS 15.1 months) and with MK-1084 monotherapy in 59 previously treated patients (ORR 27%; PFS 8.3 months).
“With 10 months of additional data since the initial report (Ann Oncol. 2025;36[Suppl 2]:S568–S569), results continue to move in the right direction for this heterogenous group, with particularly meaningful clinical activity in untreated patients with a high PD-L1 TPS,” observes Novello, noting that the latter findings support accrual to the ongoing phase III KANDLELIT-004 trial (NCT06345729). “Grade 3 increases in alanine aminotransferase and aspartate aminotransferase – 8% with dual therapy and around 11% with triple combination therapy – were the most relevant safety problem in this group, but adverse events appeared to be generally manageable,” she comments.
At a glance:
Sacher AG, et al. Updated results for MK-1084 + pembrolizumab in KRAS G12C-mutated (mut) metastatic non-small-cell lung cancer (mNSCLC) enrolled in KANDLELIT-001. European Lung Cancer Congress 2026 - Abstract 4MO
- Efficacy evaluable population:
- Dual combination: MK-1084 + pembrolizumab in untreated patients (N=98)
- Triple combination: MK-1084 + pembrolizumab + chemotherapy in untreated patients (N=46)
- Monotherapy: MK-1084 in previously treated NSCLC patients (N=59)
- ORR: Dual combination with TPS ≥50% (87%) and TPS 1–49% (55%); triple combination (65%); monotherapy (27%)
- mPFS: Dual combination (28.9 months); triple combination (15.1 months); monotherapy (8.3 months)
- Nearly two-thirds (63%) of patients receiving triple combination therapy had grade 3–4 treatment-related adverse events compared with 32% receiving dual therapy.
Naidoo J, et al. Phase II ETOP ADEPPT trial: Adagrasib in patients with KRASG12C-mutant NSCLC who are elderly or have poor performance status - Final results. European Lung Cancer Congress 2026 - Abstract 5MO
- Elderly (≥70 years and ECOG PS 0–1) (N=32); ECOG PS 2 (≥18 years) (N=34)
- cORR at 12 weeks: Elderly 10/32 (31%); ECOG PS 2 6/34 (18%)
- mPFS: Elderly 7.6 months; ECOG PS 2 2.7 months
- mOS: Elderly 9.5 months; ECOG PS 2 4.3 months