Chrono-immunotherapy may influence treatment outcomes, especially during the early phase of treatment, but its overall clinical impact remains to be clearly defined
There is mounting evidence of a biologically relevant interaction between circadian biology and anticancer immunity. The circadian timing system regulates key components of the cancer-immunity cycle, including antigen presentation, T-cell priming, immune cell trafficking and effector functioning, and the composition of the tumour microenvironment through both intrinsic cellular clocks and systemic signals (J Natl Cancer Cent. 2025;6:98–115). Importantly, this is not simply a matter of the immune response being stronger in the morning but rather reflects coordinated oscillations in immune function across the day. “In this context, the timing of immunotherapy may influence the dynamic interplay between host immunity and tumour susceptibility,” explains Dr Sara Pilotto from the University of Verona and University and Hospital Trust (AOUI) of Verona, Italy. “Delivering immunotherapy at an appropriate circadian phase could therefore modulate both the magnitude and the quality of the antitumour immune response, supporting the rationale for a chrono-immunotherapy strategy.”
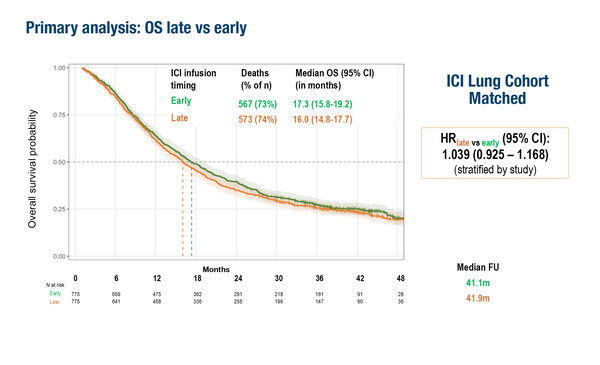
Additional evidence contributing to this evolving field was presented at the European Lung Cancer Congress 2026 (Copenhagen, 25–28 March) with the ETOP-Roche i-TIMES study, a patient-level meta-analysis of data from 8 trials in patients with lung cancer (LBA2). The study demonstrated the non-inferiority of late versus early administration of the first two ICI cycles (alone or in combination with chemotherapy) with regard to overall survival (OS).
Median OS was 16.0 months among 775 patients who received their first 2 cycles of immunotherapy after 12:00 (late group) compared with 17.3 months in 775 matched patients who received their first 2 cycles of immunotherapy before 12:00 (early group) (hazard ratio [HR] 1.039; 95% confidence interval [CI] 0.925−1.168). The upper bound of the 95% CI did not exceed the pre-specified non-inferiority limit (1.18), allowing the rejection of the hypothesis that late administration is inferior to early administration and supporting the conclusion that treatment timing within the day is unlikely to represent a clinically meaningful determinant of outcome.
These findings provide additional context to the trend observed in previous studies. A growing body of evidence from more than 30 retrospective studies conducted across multiple regions, including North and South America, Europe and Asia, evaluated the impact of time-of-day administration on immune checkpoint inhibitor (ICI) efficacy in a range of tumour types, generally suggesting a potential association between earlier treatment and improved clinical outcomes (JCO Oncol Pract. 2025;OP-25-00661). More recently, a single-centre phase III randomised trial conducted in an Asian population with advanced non-small cell lung cancer (NSCLC) reported a substantial benefit with earlier administration (Nat Med. 2026 doi:10.1038/s41591-025-04181-w); however, these findings require validation in broader clinical settings and further clarification.
"Findings from ETOP-Roche i-TIMES suggest that the impact of treatment timing on the clinical efficacy of ICIs in lung cancer may be less pronounced than previously thought. Despite leveraging data from randomised clinical trials, treatment timing itself was not randomised and some degree of residual confounding cannot be fully excluded, even with the use of matching methods,” Pilotto notes.
While the available evidence includes a biologically plausible rationale and consistent retrospective data, these new results highlight remaining uncertainties, pending prospective confirmation. Pilotto adds, “A pragmatic interpretation is that treatment scheduling may remain flexible, taking into account patient logistics, infusion unit capacity and organisation, and pharmacy workflows. Given the lack of conclusive evidence, results from ongoing trials are needed to further clarify the potential clinical implications of treatment timing.”
In this context, several ongoing trials in solid tumours are expected to provide more robust prospective data. In NSCLC specifically, the phase II TIME-NSCLC study is comparing morning administration of pembrolizumab with random scheduling in patients with metastatic disease (NCT06882174), while the phase III LungTime-C02 study is evaluating morning versus afternoon administration of neoadjuvant ICI plus platinum-based chemotherapy in patients with resectable stage II–III disease (NCT07251582), with the aim of more directly assessing the impact of timing on immune priming using pathological complete response as the primary endpoint.
“This remains an evolving area of research. Beyond the key question of its actual clinical impact, several open questions still stand, including the relevance of timing across different treatment phases, the optimal definition of early versus late administration, interindividual variability in circadian phase and the potential role of biological markers of circadian alignment. While ongoing studies may help clarify these aspects, the clinical relevance of treatment timing remains an area of active investigation and ongoing discussion,” concludes Pilotto.
At a glance:
Peters S, et al. ETOP-Roche i-TIMES: Immunotherapy timing investigation on lung cancer survival. European Lung Cancer Congress 2026 - LBA2
- N=1,550 matched pts from 8 trials (late, n=775; early, n=775)
- Median follow-up: 41.9 months (late); 41.1 months (early)
- Median OS: 16.0 months (late) vs 17.3 months (early)
- OS HRlate vs early: 1.039 (95% CI 0.925−1.168)