Early phase III trial results signal a much-needed chemotherapy-free advance with gotistobart in a setting where therapeutic choices are limited
“Treatments for patients with non-small cell lung cancer (NSCLC) when their disease progresses after immunotherapy is a huge unmet need as there are currently very few options available,” explains Dr Marina Garassino of the University of Chicago, IL, USA. “Because these patients do not harbour actionable alterations, they are excluded from being offered targeted therapies. While in general they respond to immunotherapy or a combination of immunotherapy and chemotherapy, their outcomes remain very poor.”
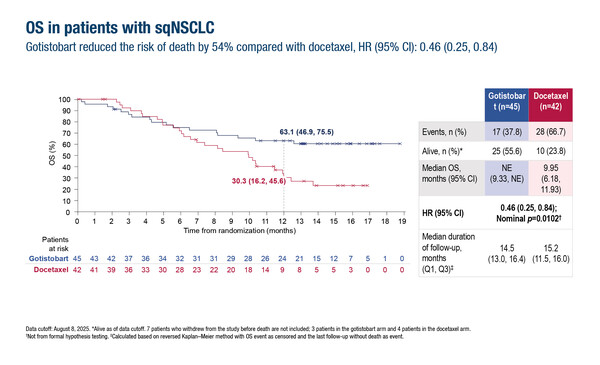
In this setting, results from stage 1 of the PRESERVE-003 trial presented at the European Lung Cancer Congress 2026 (Copenhagen, 25–28 March) show promise in expanding therapeutic options for patients with metastatic squamous NSCLC who progressed on anti-PD-(L)1 therapy as gotistobart improved the primary endpoint of overall survival (OS) compared with docetaxel (Abstract 3O). In the study, at median follow-up of around 15 months, median OS was not reached in 45 patients treated with gotistobart, a novel target-preserving anti-CTLA-4 antibody that acts via selective depletion of regulatory T-cells in the tumour microenvironment (J Clin Oncol. 2023;41(16_suppl.):9024). However, median OS was 9.95 months in 42 patients treated with docetaxel (hazard ratio [HR] 0.46; 95% confidence interval [CI] 0.25–0.84; nominal p=0.0102). The 12-month progression-free survival rate was 25.2% with gotistobart versus 0% with docetaxel (HR 0.69; 95% CI 0.42–1.13). The confirmed objective response rate was 20.0% with gotistobart and 4.8% with docetaxel, with a median duration of response of 11.0 months and 3.8 months, respectively.
A key finding in this study is the long duration of response to gotistobart, which may be central to the benefit seen with this agent,” notes Garassino. “However, a crucial missing element is the lack of available biomarkers to signal a response to anti-CTLA-4 therapy.”
Researchers reported grade ≥3 treatment-related adverse events (TRAEs) that were experienced by 42.2% of patients in the gotistobart arm and 48.8% of patients in the docetaxel arm, most commonly colitis (8.9%) and increased alanine aminotransferase (6.7%) with gotistobart. In total, 13.3% of patients experienced TRAEs leading to discontinuation in the gotistobart arm compared with 4.9% in the docetaxel arm.
Initially, patients with squamous and non-squamous NSCLC were enrolled in stage 1 of PRESERVE-003, but only patients with squamous NSCLC are included in the ongoing pivotal stage 2 part of the trial (NCT05671510). “This will help determine the potential role of gotistobart in this setting, but what is also needed is information on the use of gotistobart after progression on prior anti-CTLA-4 antibody therapy, as well as greater characterisation of the patients responding to this type of therapy,” concludes Garassino.
Programme details
He K, et al. Anti-tumor activity of gotistobart compared to docetaxel in patients with metastatic squamous non-small cell lung cancer (sqNSCLC) progressing on PD-(L)1 inhibitors: Stage 1 PRESERVE-003 phase III trial. European Lung Cancer Congress 2026 - Abstract 3O