From geriatric assessment to artificial intelligence, an ESMO/SIOG paper highlights effective strategies to improve the representation of older patients in clinical trials
The underrepresentation of older people in cancer research has become an increasingly urgent issue in ensuring high-quality care to all patients. In 2020, 64% of new cancer cases worldwide occurred in individuals aged 60 or above, and cancer incidence among the older population is expected to reach 20.7 million new cases by 2040 (J Natl Cancer Cent. 2024 May 9;4(3):223–232). Despite the global cancer patient population aging, most of what we know about the risks and benefits of cancer therapeutics is based on clinical trials conducted in younger, fitter and healthier patients, and treatment strategies do not adequately address the diversity, priorities and expectations of older patients (CA Cancer J Clin. 2020 Oct 1;71(1):78–92).
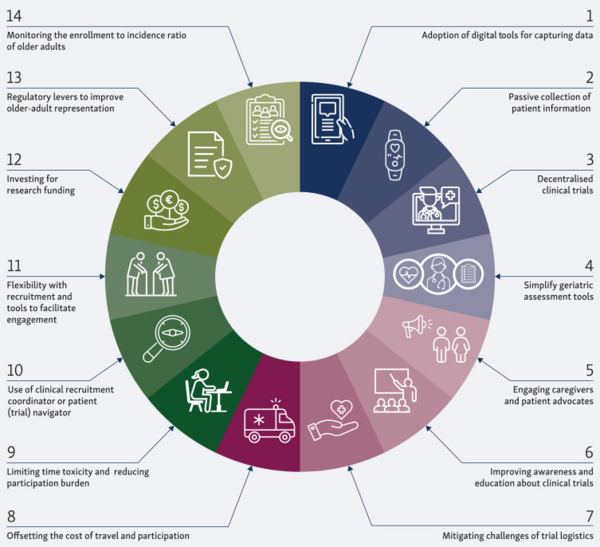
"While clinical trials typically focus on traditional outcomes such as treatment efficacy and survival, older adults often value the quality of time as much as the quantity of time,” explains Dr Anna Mislang from the Department of Medical Oncology, Flinders Centre for Innovation in Cancer, Bedford Park, South Australia, Australia. "Priorities such as quality of life and maintaining functional independence are frequently overlooked or underreported, but it is vital to define research endpoints that reflect the specific needs and preferences of this patient population.” Mislang is part of the ESMO/SIOG Cancer in the Elderly Working Group which has identified strategies to improve clinical trial designs and endpoints by better integrating older patients’ needs, now available on ESMO Open (Articles in Press, February 22, 2026).
Adapting trial design to older patients
When designing clinical trials specifically for older cancer patients, several critical factors need to be considered, including sensory limitations or cognitive impairments who may result in additional time constraints for research teams. “Enrolling an older adult often requires more comprehensive assessments, including evaluations of functional status and patient priorities, which can be difficult to accommodate when time and resources are limited,” adds Dr Capucine Baldini, from the Drug Development Department (DITEP) at the Gustave Roussy, Villejuif, France, who authored the paper with Mislang. “Also, from the clinician’s perspective, age-related bias can play a significant role: physicians may be concerned about potential toxicities of new treatments and hesitant to add additional burden to older patients.”
Despite these and other barriers, incorporating Geriatric Assessment and Management (GAM) into care pathways, as well as using lower or stepwise dose interventions or stratification based on Geriatric Assessment (GA), which may improve treatment tolerance, appear to be effective approaches to enhance clinical trial design and increase older patients’ inclusion and participation. “Shifting the focus toward quality of life and functional independence transforms treatment decisions from disease-centred to patient-centred. While integrating these endpoints is complex, it can be achieved through GA-guided adaitive trial designs,” continues Mislang. “Without systemic shifts in how trials are designed and regulated, clinician and patient-related barriers will remain secondary to a framework that inherently excludes the older participants.”
The pivotal role of the caregiver
According to the ESMO/SIOG international experts, recognising the caregiver’s role in clinical trial design can enhance the overall care experience for both patients and their families. Communication is key to this aspect, and the implementation of GA in clinical practice offers a way to improve patient and caregiver satisfaction as well as the number and quality of conversations about aging-related concerns compared to those who received usual care, as demonstrated in a cluster-randomised trial (JAMA Oncol. 2020 Feb 1;6(2):196-204). “During the GA, there is dedicated time to discuss the patient’s priorities and to explain the clinical trial in greater detail, including its objectives and the potential benefits for the patient and the broader community. Ensuring a clear understanding of the clinical trial is critical, and both patients and caregivers must fully understand the trial design and requirements,” clarifies Baldini. “Although cognitive impairment can be a barrier, decision-support tools are now available and can be provided to both patients and caregivers to support comprehension. We use these tools in practice and highlight them in the paper, as they are an important component of the informed-consent process, requiring adequate time and consideration.”
Finally, attention must be given also to the patient-caregiver dyadic relationship. “Frequently, older patients do not want to burden their caregivers. When asked about limitations to participation in clinical trials, in fact, this concern is often among the first mentioned,” she continues. “Aligning the priorities of both patients and caregivers is therefore essential to improving enrollment.”
AI as a tool to bridge underrepresentation
Conducting clinical trials can be difficult when patient engagement and retention is low, as is the case in oncology research involving older adults. However, artificial intelligence (AI) now offers promising solutions. One such is the creation of synthetic control arms, by using existing data from multiple sources, enabling research to be conducted when traditional control arm is difficult, unethical, or impractical.
“Additionally, AI-driven screening platforms can potentially analyse electronic health records to identify eligible older participants who might otherwise be overlooked by manual review. Decentralised clinical trials supported by wearable sensors and remote monitoring, also reduce the physical and geographic burdens of participation for those with limited mobility or living remotely,” states Mislang echoing some proposals made in the paper.
Despite these promising opportunities, the persistence of a digital divide in the general population may limit their impact on cancer research. “To be effective, all these tools must be co-designed with older adults to ensure they are accessible, and researchers must ensure that the underlying AI models do not inadvertently perpetuate ageist biases by using unrepresentative data,” she suggests.
Industry-driven research remains limited
Almost 15 years have passed since the European Medicines Agency issued guidance to better address the needs of older people in clinical research. While the oncology community has become more aware of the underrepresentation of these patients in clinical trials, tangible progress has lagged behind. A study investigating age disparities in oncology clinical trials reported that among 7,747 cancer trials registered in the period 2008–2021, only 1.5% of them included patients aged 60 years or more (BMC Cancer. 2024 Jan 2;24(1):30). For the ESMO/SIOG working group, funding is a major obstacle. “This is particularly true in industry-sponsored trials compared to academic trials,” highlights Baldini. “Engaging with industry to improve the representation of older adults in clinical trials requires an open and ongoing dialogue.”
This is an area where the joint group of experts has been actively engaged, particularly in collaboration with regulatory authorities, which could be a game-changer. “For instance, regulators could require the enrollment of a minimum proportion of older adults in clinical trials as a condition for drug approval, ensuring that trial results are externally valid and applicable to the broader patient population,” concludes Baldini. “This is a key point we wanted to emphasise in our work: while progress is incremental, stronger regulatory frameworks are essential to drive meaningful and lasting improvements in the representation of older adults in clinical research.”
Geriatric assessment and management (GAM) is essential for delivering personalised, patient-centered cancer care in older adults, as highlighted in a position paper of the ESMO/SIOG Cancer in Elderly Working Group in 2024. Geriatric assessment (GA) evaluates multiple domains influencing prognosis and treatment decisions in older adults, including functional status, comorbidities, cognition, psychological health, social support, nutrition, and medications. Assessing all relevant domains is more important than using multiple tools within each domain, and tool selection should reflect local resources and expertise. Experts encourage to use GA to directly inform clinical management, with reassessment over time or at disease progression enabling adaptive treatment and supportive care decisions.