An easy-to-use method to detect dysbiosis may help guide selection of cancer treatments and microbiota-centred interventions
The influence of the gut microbiome on response to immunotherapy is now well established; however, tools to help detect dysbiosis are lacking and no consensus exists on a gut fingerprint predicting immunoresistance. In a study presented at ESMO Asia Congress 2022 (Singapore, 2–4 December), a test for gut microbiota was shown to have predictive value for overall survival (OS) after immune checkpoint inhibitor (ICI) therapy in patients with non-small-cell lung cancer (NSCLC) or renal-cell carcinoma (RCC) (Abstract 259MO). It has previously been shown that primary resistance to ICIs may be attributed to abnormal gut microbiome composition due to tumourigenesis, with antibiotics also playing a role in inhibiting the clinical benefit of treatments in patients with advanced cancer (Science. 2018;359:91–97).
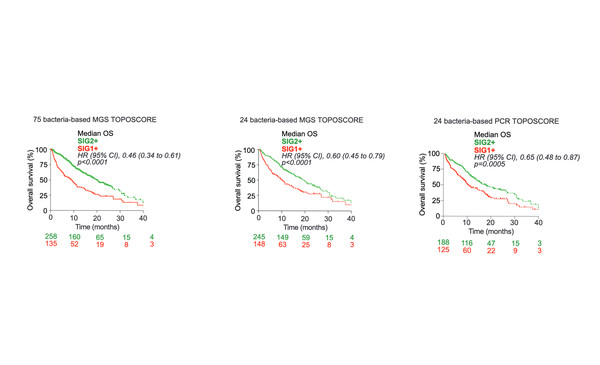
The ONCOBIOTICS study was conducted to define metagenomic signatures associated with cancer immunotherapy outcomes. In a discovery cohort of 245 patients with NSCLC receiving an ICI, shotgun metagenomics sequencing (MGS) followed by microbial genomic profiling was used on faecal samples and only microbial species having a prevalence of ≥2.5% were considered for the analysis. Two distinct microbial consortia or species interacting groups (SIG) were identified. One contained 40 harmful species (termed ‘SIG1’) and the other contained 34 beneficial species (termed ‘SIG2’), which related to overall survival (OS) of less than 12 months (non-responders) or more than 12 months (responders). A monodimensional score, the TOPOSCORE, was then computed based on the SIG1/SIG2 ratio, combined with the relative abundance of Akkermansia muciniphila (Akk), which is known to predict clinical response to PD-1 blockade from the group’s previous studies (Nat Med. 2022;28:315–324).
The TOPOSCORE performed well in the discovery cohort, with sensitivity, specificity, and positive and negative predictive values of 79%, 50%, 62% and 69%, respectively. Furthermore, multivariate analysis revealed that the TOPOSCORE was an independent prognostic factor, with a hazard ratio of 0.54 (95% confidence interval [CI] 0.40–0.72; p<0.001) comparing SIG1 and low Akk abundance versus SIG2 and high/0 Akk abundance. TOPOSCORE was then found to predict OS in a validation cohort of 148 patients with NSCLC and a prospective cohort of patients with NSCLC (n=61) or RCC (n=83), and it outperformed the International Metastatic RCC Database Consortium (IMDC) risk model. To increase ease of use, a PCR-based TOPOSCORE test was developed from the MGS-based TOPOSCORE and these matched each other in terms of prediction of OS benefit.
Based on the results presented, TOPOSCORE may help stratify patient populations amenable to ICI, to ascribe their resistance to intestinal dysbiosis and to better guide cancer treatments and compensatory microbiota-centred interventions.
Abstract discussed:
Derosa L, et al. A predictive score of cancer immunotherapy responses based on ecological analysis of gut microbiota. ESMO Asia Congress 2022, Abstract 259MO
Mini Oral Session: Developmental and precision medicine 02.12.2022, h. 14:30 – 16:00, Summit 2. Also watch the session on the Congress virtual platform.