Survival benefit with first-line pembrolizumab plus chemotherapy in persistent, recurrent or metastatic cervical cancer
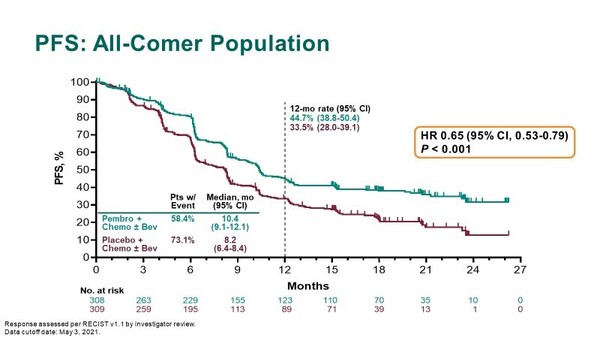
At Presidential Symposium I today, a statistically significant 33% improvement in overall survival (OS) was reported with first-line combination therapy with pembrolizumab versus placebo (24.4 months versus 16.5 months, respectively; hazard ratio [HR] 0.67; 95% confidence interval [CI] 0.54–0.84; p<0.001) in 617 patients with recurrent or metastatic cervical cancer who did not previously receive systemic chemotherapy and were no longer candidates for curative treatments such as surgery and/or radiation (LBA2_PR). The pembrolizumab combination also led to significantly longer progression-free survival (PFS) (10.4 months versus 8.2 months, respectively; HR 0.65; 95% CI 0.53–0.79; p<0.001) (Figure).
Whether immunotherapy could be used even earlier in cervical cancer, in localised disease, is an important follow-up question.
This large randomised international phase III trial, with a well-designed control arm, provides a high level of evidence for the efficacy of pembrolizumab plus chemotherapy for prolonging survival in the first-line setting – these data will change medical practice,” says Prof. Isabelle Ray-Coquard from Centre Léon Bèrard, Université Claude Bernard Lyon I, Lyon, France. She continues, “The large survival improvements observed are clearly needed in this vulnerable population who currently are underserved. Cervical cancer has been the ‘poor relation’ in gynae-oncology and in oncology in general, but this study provides further support for immunotherapy as the new cornerstone of its treatment.” Ray-Coquard thinks that the utility of this regimen could be explored further: “Whether immunotherapy could be used even earlier in cervical cancer, in localised disease, is an important follow-up question.”
Across subgroups defined by PD-L1 combined positive score (CPS), Ray-Coquard points out that OS HRs were similar for pembrolizumab versus placebo in all comers (HR 0.67), those with PD-L1 CPS ≥1 (HR 0.64) and in those with PD-L1 CPS ≥10 (HR 0.61), but data on effects in patients with PD-L1 CPS 0 would help to complete the picture.
In KEYNOTE-826, patients received platinum-based chemotherapy (paclitaxel with cisplatin or carboplatin), with bevacizumab (63.6%) or without, at the investigators’ discretion. The benefits of the pembrolizumab combination were seen regardless of bevacizumab use. “Understanding how best to combine bevacizumab with pembrolizumab in this setting should now be explored,” says Ray-Coquard. “The trial was not randomised for bevacizumab and therefore further studies are needed. Whether we need chemotherapy at all, particularly in patients with PD-L1 CPS ≥10, is up for debate and requires clinical investigation.”
There appeared to be no unexpected safety signals with the pembrolizumab combination. From the data presented, the adverse event profile, including neutropenia and anaemia, appeared manageable and consistent with previous studies. Despite this, and as a note of caution, Ray-Coquard adds, “Pembrolizumab treatment is of longer duration than chemotherapy alone and patients will need to be supported to understand the benefits of treatment over 2 years.”
Colombo N et al. Pembrolizumab plus chemotherapy versus placebo plus chemotherapy for persistent, recurrent, or metastatic cervical cancer: randomized, double-blind, phase 3 KEYNOTE-826 study. ESMO Congress 2021, Abstract LBA2
Presidential symposium 1, 18.9.2021, h. 15:35 – 15:50, Channel 1
Simultaneous publication in The New England Journal of Medicine, DOI: 10.1056/NEJMoa2112435