A pathological complete response-guided approach enables treatment optimisation in a selected population with HER2-positive breast cancer in the PHERGain-2 study
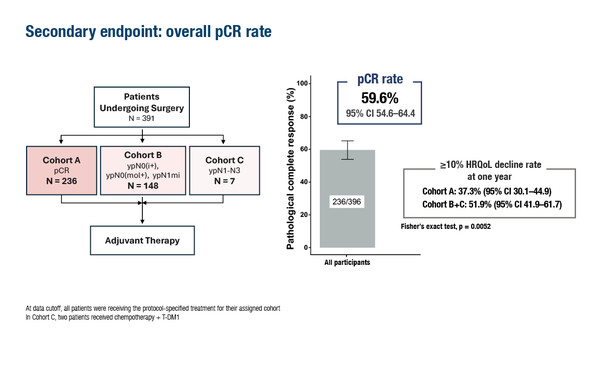
“Current treatments for HER2-positive breast cancer are extremely effective, but it is important that patients with a high likelihood of cure only receive systemic treatment necessary to maximise benefit while minimising toxicity,” highlights Dr Giampaolo Bianchini from IRCCS Ospedale San Raffaele, Milan, Italy, discussing the remarkable results of the PHERGain-2 study presented at the ESMO Breast Cancer 2026 congress (Berlin, 6–8 May). Improving patient quality of life without compromising clinical outcome is today a priority in oncology, and treatment optimisation based on chemotherapy-free regimens continues to prove to be a promising approach. In the study, patients who achieved pathological complete response (pCR; 59.6%) at 1 year following treatment with neoadjuvant trastuzumab–pertuzumab and continued on a chemotherapy-free regimen had a lower incidence of decline in health-related quality of life (HRQoL) of ≥10% compared with patients who did not achieve pCR and continued on ado-trastuzumab emtansine (T-DM1) with/without chemotherapy (37.3% versus 51.9%, respectively) (Abstract 214O).
The PHERGain-2 multicentre, open-label, phase II study aims to optimise treatment and improve quality of life for patients with small (5–30 mm), node-negative, early-stage HER2-positive (immunohistochemistry [IHC] 3+) breast cancer using a de-escalation strategy based on pCR. Overall, 391 of 396 enrolled patients underwent surgery following neoadjuvant treatment. Adjuvant therapy post-surgery was adapted according to response and patients were treated in 3 cohorts: those with pCR continued to receive trastuzumab–pertuzumab (cohort A; n=236); patients with residual invasive disease in the breast and with isolated tumour cells or micrometastases received T-DM1 (cohort B; n=148); and those with 1–3 positive lymph nodes could receive optional adjuvant chemotherapy before T-DM1 (cohort C; n=7). The 72.7% of the study population with hormone receptor (HR)-positive tumours also received endocrine neoadjuvant and adjuvant therapy.
The study builds on the concept previously explored in the initial PHERGain trial (Lancet. 2024;403:1649–1659). In this study, early metabolic imaging response and pCR to inform and individualise subsequent treatment in patients with stage I–IIIA invasive, operable HER2-positive breast cancer first introduced. FDG-PET response after 2 cycles (6 weeks) of chemotherapy-free neoadjuvant trastuzumab plus pertuzumab (with/without endocrine therapy) guided subsequent neoadjuvant therapy with continuation of this regimen in responders or starting chemotherapy plus trastuzumab and pertuzumab in non-responders; pCR following surgery guided choice of adjuvant therapy. This approach enabled approximately one-third of patients to omit chemotherapy and resulted in a 3-year invasive disease-free survival of 94.8%.
“The PHERGain-2 trial represents a further advance in that it includes a highly selected, favourable-risk population with small, node-negative tumours, and only those with cancers that were HER2 3+ by IHC, which have greater sensitivity to HER2-targeted agents,” notes Bianchini. All patients received the same neoadjuvant therapy, but pCR following surgery allowed tailoring of adjuvant therapy. At surgery, only 7 patients (1.8%) had evidence of disease in their lymph nodes, and only 2 patients (0.5%) discontinued the study due to disease progression. “This is very reassuring for patients who may have concerns relating to the efficacy of treatment in the absence of chemotherapy,” he adds. Most study treatment-related adverse events (TRAEs) were mild to moderate in severity (grade ≥3 TRAEs were reported in 5.3% of patients and serious TRAEs in 1.0% of patients).
According to Bianchini, the overall study results, although immature due to the lack of event-free survival data, are potentially practice-changing: “Most patients were able to avoid cytotoxic chemotherapy or antibody–drug conjugate therapy, and this was accompanied by preservation of HRQoL for the majority,” he says. “Also, the treatments are already available in the clinic and could be implemented directly once data on longer-term efficacy are reported.”
Over the last decade, several trials have accumulated evidence on the feasibility of treatment optimisation in selected populations of patients with small, node-negative HER2-positive breast cancer. These include: the APT study of adjuvant paclitaxel plus trastuzumab demonstrating excellent long-term outcome despite reducing chemotherapy exposure (N Engl J Med. 2015;372:134–141; Lancet Oncol. 2023;24:273–285); the ATEMPT trial in which adjuvant paclitaxel plus trastuzumab was compared with 17 cycles of T-DM1 to determine whether toxicity could be reduced versus the chemotherapy-containing regimen (J Clin Oncol. 2021;39:2375–2385; J Clin Oncol. 2024;42:3652–3665); the ongoing ATEMPT 2.0 trial (NCT04893109) investigating whether a shorter (6-cycle) course of adjuvant T-DM1 followed by standard trastuzumab can further reduce toxicity; and the ADEPT study (NCT04569747) evaluating a chemotherapy-free regimen of adjuvant trastuzumab plus pertuzumab and endocrine therapy in patients with HER2-positive, HR-positive disease.
The future of research into treatment optimisation will depend on validated biomarkers. “The implementation of a biomarker strategy could further help to select patients most likely to benefit from a pCR-guided approach, while data from the ongoing ATEMPT 2.0 trial may contribute to a clearer understanding of the optimal duration of T-DM1,” concludes Bianchini.
Programme details
Llombart Cussac A, et al. Chemotherapy-free, pathological complete response (pCR)-guided strategy with trastuzumab-pertuzumab (HP) and T-DM1 in HER2+ early breast cancer (EBC): PHERGain-2. ESMO Breast Cancer 2026. Abstract 214O