The first prospective phase II trials evaluating TOPO1 ADC after prior TOPO1 ADC therapy confirm the emergence of cross-resistance
Whilst antibody–drug conjugates (ADCs) have revolutionised the management of metastatic breast cancer (MBC), showing dramatic improvements in key efficacy outcomes across several subtypes of the disease (Ther Adv Med Oncol. 2025;17:17588359251396656), data guiding their optimal sequencing remain limited. In two prospective studies presented at the ESMO Breast Cancer 2026 congress (Berlin, 6–8 May), limited activity of patritumab deruxtecan (HER3-DXd) and sacituzumab govitecan was observed in patients previously treated with topoisomerase 1 (TOPO1) inhibitor-based ADCs. Despite targeting different antigens, both agents share a TOP1 payload backbone and the consistently low efficacy reported across these studies suggests that switching the ADC target alone may be insufficient to overcome resistance.
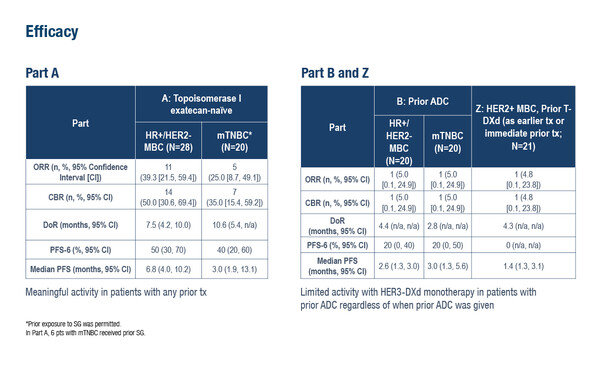
In a first phase II study presented at the congress, efficacy of HER3-DXd was evaluated across 3 patient cohorts: metastatic HR-positive/HER2-negative or triple negative breast cancer (TNBC) naïve to TOPO1 exatecan ADCs (Part A); the same MBC subtypes but with prior exposure to TOPO1 ADCs (trastuzumab deruxtecan [T-DXd] or datopotamab deruxtecan [Dato-DXd] and/or sacituzumab govitecan; Part B); and HER2-positive MBC with progression on prior T-DXd (Part Z) (Abstract 422RO).
ADC-naïve patients (Part A) derived clinically meaningful benefit from HER3-DXd monotherapy; outcomes were improved in patients with HR-positive/HER2-negative MBC compared with those with TNBC: objective response rate (ORR) 39.3% versus 25.0%, median progression-free survival (PFS) 6.8 months versus 3.0 months and PFS at 6 months 50% versus 40%. Limited activity was observed in patients with prior TOPO1 ADC exposure from study Parts B and Z, with no differences in efficacy between tumour types.
Similarly poor outcomes were reported in the phase II SATEEN trial of sacituzumab govitecan plus trastuzumab in patients with HER2-positive MBC after prior T-DXd (LBA4). The small study population (n=27) had been heavily pre-treated, with a median of 5 (range 1–17) prior lines of HER2-directed therapy for metastatic disease. The ORR was 3.7%, with only 1 confirmed partial response. Median PFS was 2.3 months and median overall survival was 9.2 months. Providing the first prospective data for sacituzumab govitecan after prior T-DXd in HER2-positive MBC, the findings suggest limited benefit from sequential TOPO1 inhibitor-based ADCs in this setting.
“The ADCs evaluated in each study have a common cytotoxic TOPO1 inhibitor payload despite different monoclonal antibody targets (HER3 for HER3-DXd, TROP2 for sacituzumab govitecan and HER2 for T-DXd),” notes Dr Thomas Grinda from Gustave Roussy, Villejuif, France. He cautions that the prospective data are derived from relatively small patient cohorts and therefore cannot be considered definitive evidence against the clinical benefit of sequential TOPO1 inhibitor-based ADCs in MBC. “Nevertheless, these findings provide a consistent signal that switching the ADC target alone is unlikely to overcome resistance in many patients when the payload remains the same. This highlights the potential role of payload-associated cross-resistance and underscores the need for alternative sequencing strategies and novel therapeutic approaches to improve outcomes,” he adds.
The findings presented in Berlin are consistent with real-world studies that have reported limited efficacy in patients receiving TOPO1 ADC after prior TOPO1 ADC (NPJ Breast Cancer. 2025;11:34; J Clin Oncol. 2024;42(16_Suppl):1085).
Data from translational studies have suggested potential mechanisms of payload-associated resistance, including mutations in payload target genes such as TOP1, efflux transporter upregulation, and activation of bypass survival pathways (NPJ Breast Cancer. 2025;11:34; Cancer Discov. 2021;11:2436–2445; Drug Resist Updat. 2026;85:101353).
“This remains a key challenge in MBC where most available ADCs rely on a TOPO1 inhibitor payload,” notes Grinda. Ongoing biomarker analyses are expected to further clarify the mechanisms underlying resistance to TOPO1-based ADCs and to inform more effective post-ADC treatment strategies (Clin Cancer Res. 2025;31:1824–1826; Drug Resist Updat. 2026;85:101353). “I anticipate that biomarker-driven sequencing will become increasingly important,” he adds. “Repeat biopsy, circulating tumour DNA, and other dynamic biomarkers could help identify emerging resistance mechanisms – such as TOP1 alterations or antigen loss – and support a more rational selection of subsequent therapies following ADC exposure,” Grinda suggests.
Several strategies are currently being developed to overcome ADC cross-resistance. Combination approaches are among the most advanced, with ADCs being paired with immune checkpoint inhibitors and targeted therapies to exploit complementary vulnerabilities. Grinda notes that combinations with PARP inhibitors are particularly appealing in the context of TOPO1-based ADCs, as PARP plays a central role in repairing TOPO1 inhibitor-induced DNA damage. Inhibiting PARP can therefore enhance cytotoxicity through a synthetic lethality mechanism. Beyond combinations, other approaches focus on payload diversification, with the development of alternative cytotoxic agents designed to overcome mechanisms such as efflux-mediated resistance. In parallel, next-generation ADC formats are emerging, including bispecific ADCs and immune-stimulating antibody conjugates, which aim to improve tumour targeting and engage additional antitumour mechanisms. Finally, advances in ADC engineering, such as novel linker technologies and site-specific conjugation strategies, may enhance payload delivery and intracellular release, thereby reducing resistance linked to suboptimal drug distribution or processing (NPJ Breast Cancer. 2025;11:102; Nat Rev Clin Oncol. 2024;21:203–223).
At glance
Hamilton EP, et al. A phase 2 study of patritumab deruxtecan (HER3-DXd) in patients (pts) with metastatic breast cancer (MBC). ESMO Breast Cancer 2026 - Abstract 422RO
- Part A: Metastatic HR+/HER2– or TNBC, ADC-naïve (n=60)
- Part B: Metastatic HR+/HER2– or TNBC, prior treatment with TOPO1 ADC (n=40)
- Part Z: HER2+ MBC and prior T-DXd (n=21)
- For patients in Part A (HR+/HER2– MBC and mTNBC), Part B (HR+/HER2– MBC and mTNBC) and Part Z:
- ORR: 39% and 25%, 5% and 5%, 5%
- DOR: 7.5 months and 10.6 months, 4.4 months and 2.8 months, 4.3 months
- PFS at 6 months: 50% and 40%, 20% and 20%, 0%
- Median PFS: 6.8 months and 3.0 months, 2.6 months and 3.0 months, 1.4 months
- Grade ≥3 AEs: 51% overall
- Treatment discontinuation (Part A, B and Z): 18%, 5% and 0%
- Deaths due to AEs (Part A, B and Z): n=2 (Pneumocystis jirovecii pneumonia and ILD), n=1 (respiratory failure due to ILD progression) and 0
Tarantino P, et al. Efficacy and safety of sacituzumab govitecan (SG) plus trastuzumab in patients with HER2+ metastatic breast cancer after prior trastuzumab deruxtecan (T-DXd): Results from the phase II SATEEN trial. ESMO Breast Cancer 2026 - LBA4
- N=27 (stage 1)
- Median number of prior lines of HER2-directed therapy for MBC: 5 (range 1–17)
- ORR: 3.7% (1 confirmed PR)
- Median PFS: 2.3 months
- Median OS: 9.2 months
- Grade 5 TRAE: n=1 (neutropenic sepsis with multiorgan failure)
- Trial did not meet primary endpoint (ORR) and was stopped at the interim analysis for futility