Encouraging results with giredestrant support the future use of SERD-based endocrine strategies in this setting
The selective oestrogen receptor degrader (SERD), giredestrant, with or without the luteinising hormone-releasing hormone (LHRH) analogue, triptorelin has been associated with robust anti-cancer activity in the PREcoopERA window-of-opportunity trial (LBA2). “The findings from PREcoopERA add to our understanding of how we can optimally provide endocrine therapy to younger patients with oestrogen receptor (ER)-positive/HER2-negative breast cancer – a population with unique needs and medical considerations,” highlights Dr Erica Mayer from the Dana-Farber Cancer Institute and Harvard Medical School, Boston, MA, USA. The data were presented at the ESMO Breast Cancer 2026 congress (Berlin, 6–8 May).
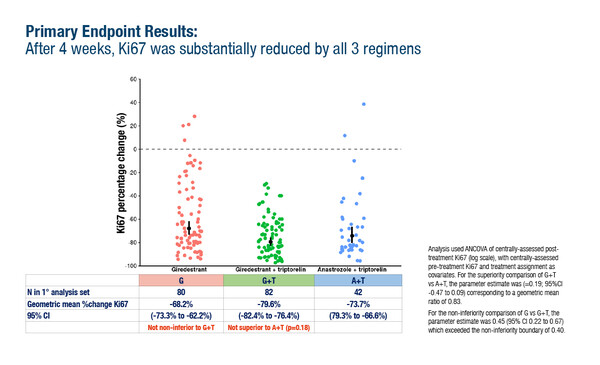
In the study, premenopausal patients with untreated ER-positive/HER2-negative, stage I–III breast cancer with Ki67 >10% received 28 days of pre-operative treatment on one of three arms: giredestrant plus triptorelin, the aromatase inhibitor anastrozole plus triptorelin, or giredestrant monotherapy. The primary endpoint was centrally assessed change in Ki67 between baseline and post-treatment biopsy or surgery. The trial tested two hypotheses: that giredestrant plus triptorelin shows superiority to anastrozole plus triptorelin, and that giredestrant shows non-inferiority to giredestrant plus triptorelin. In the primary analysis population (N=204), there were marked reductions in Ki67 in all arms versus baseline, with geometric mean changes of -79.6% (95% confidence interval [CI] -82.4, -76.4) with giredestrant plus triptorelin, -73.7% (95% CI -79.3, -66.6) with anastrozole plus triptorelin (log difference -0.19 [95% CI -0.47, 0.09]; p=0.18), and -68.2% (95% CI -73.3, -62.2) with giredestrant alone (log difference versus giredestrant plus triptorelin: 0.45 [95% CI 0.22, 0.67]). No significant difference in activity was observed between the two LHRH-containing arms, and the giredestrant arm, while active, was not non-inferior to giredestrant plus triptorelin.
Oral SERDs are an exciting class of endocrine treatment with proven robust efficacy and tolerability in the metastatic setting. Giredestrant and the approved oral SERD imlunestrant were both associated with significant progression-free survival (PFS) benefits versus standard of care when used in combination regimens in ER-positive/HER2-negative advanced breast cancer: giredestrant with everolimus in the phase III evERA trial (Ann Oncol. 2025;36(Suppl 2):S1561–S1562), and imlunestrant with or without abemaciclib in the phase III EMBER-3 trial (Ann Oncol. 2026;37:532–543). “There is growing interest in moving these drugs into earlier settings following recent positive data,” says Mayer, mentioning positive results from recent studies such as the phase III lidERA trial and the phase II SOLTI-2104-PremiÈRe study (Clin Cancer Res. 2026;32(4_Suppl):GS1-10; Clin Cancer Res. 2026;32(4_Suppl):PD10-01).
“We are seeing an increasing incidence of breast cancer in younger, premenopausal patients for whom specialised breast cancer treatment may be needed. The addition of ovarian function suppression (OFS) induces a postmenopausal state that can be associated with side-effects that may impair quality of life and reduce adherence with oral medications. Thus, for younger patients, it would be advantageous if effective endocrine therapy could be provided without the need for OFS.” However, results from the PREcoopERA trial suggests giredestrant has greater activity in the presence of OFS. The findings align with data from the phase II SOLTI-2104-PremiÈRe study (Clin Cancer Res. 2026;32(4_Suppl):PD10-01). “We are unable to suggest any changes to current practice based on these data, and further research will be needed to understand whether there is a subgroup for whom LHRH treatment may be omitted from oral SERD therapy without impairing efficacy,” she surmises.
Overall, treatment was well tolerated in the PREcoopERA study, with few grade 3 adverse events (AEs) and no grade 4–5 AEs reported. “It would be interesting to know the oestradiol levels in these patients, as oral SERDs may raise oestradiol and this could be associated with side-effects such as ovarian cysts,” she says. Ovarian cysts were reported in 2 patients in the giredestrant monotherapy arm of the PREcoopERA trial.
The last few years have seen numerous clinical investigations into novel endocrine-based therapies for breast cancer, and at least four oral SERDs have now been either approved by the U.S. Food and Drug Administration (elacestrant and imlunestrant) or have registration data (giredestrant and camizestrant). Favourable clinical findings have also been reported with other novel, next-generation ER-targeting agents, including PFS benefit with vepdegestrant, an oral proteolysis-targeting chimera ER degrader, in a phase III study of patients with HR-positive/HER2-negative advanced breast cancer and ESR1 mutations (N Engl J Med. 2025;393:556–568). Antitumour activity has also been demonstrated with palazestrant, a novel oral complete ER antagonist and SERD in a phase I/II study of heavily pre-treated HR-positive/HER2-negative patients with wild-type and ESR1-mutated advanced breast cancer (Breast Cancer Res. 2025;27:119). “It is hoped that this new wave of novel endocrine agents will improve efficacy and tolerability for our younger patients with early breast cancer,” Mayer concludes.
Programme details
Munzoni E, et al. A window-of-opportunity (WOO) trial of giredestrant +/- LHRH analogue vs anastrozole + LHRHa in premenopausal patients with ER+/HER2- early breast cancer: PREcoopERA. ESMO Breast Cancer 2026 - LBA2