A new ESMO consensus-driven document provides definitions, formats and minimum required data to uniform enumeration and reporting of prior lines of therapy across solid tumours
There is an intended treatment sequence outlined during initial care planning, and then there is reality. Many patients with cancer undergo several adjustments in their care pathways, due to the evolving biology of the disease, response to treatment, and individual needs. In this scenario, accurately enumerating prior lines of therapy (LoT) is not merely an administrative exercise – it is crucial to treatment selection, clinical auditing, trial eligibility, adherence to regulatory indications, health technology assessments, and reimbursement (Br J Cancer. 2021 Apr 13;125(2):155–163 ). An apparently straightforward task is, however, far more complex in real-world settings.
Why is accurately enumerating lines of therapy important?
“When one component of a multi-agent regimen is discontinued without evidence of progression, this would generally not be considered a change in LoT. Yet if a new drug is added to an ongoing regimen in the absence of progression, many clinicians would record this as a new LoT,” explains Dr Miriam Koopman, University Medical Center Utrecht, The Netherlands, and Chair of the ESMO AI & Digital Oncology Committee. Discrepancies exist among oncologists in what they consider a ‘line’. “Similarly, when a drug within a regimen is substituted due to intolerance – whether with an agent of similar or different mechanism of action – opinion is divided. The treatment holiday is another area of contention: when a patient responding to treatment is given a break and later restarts the same regimen, whether this constitutes a continuation or a new LoT depends on individual interpretation.”
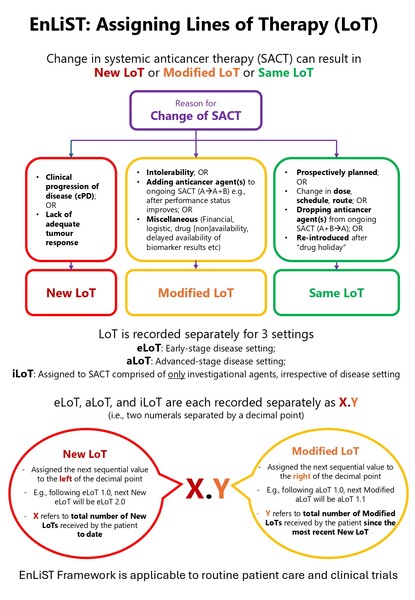
Newly published in Annals of Oncology, the ESMO Adaptation of Lines of Systemic Therapy (EnLiST) is the first consensus-driven effort to address this ambiguity, providing core definitions, a standard format for reporting LoT, the minimum required data for reporting LoT, and detailed guidelines for uniform enumeration of LoT applicable to all solid tumours (Ann. Oncol 2026, Special article, February 18). It further expands the framework proposed in 2021 by Dr Kamal Saini, Department of Medical Oncology, Cambridge University Hospitals NHS Foundation Trust, UK, and Prof. Chris Twelves, University of Leeds and Leeds Teaching Hospitals Trust, UK. “ESMO developed and refined the initial framework proposed by us (Saini and Twelves) into a consensus-driven standard guiding principle framework. ESMO brought international credibility and authority, and a broad panel of experts and stakeholders including clinicians, trialists, and regulators and HTA bodies,” says Saini.
Reducing variability in clinical trials
“Lack of standard methodology in enumerating LoT can result in inconsistent clinical decision-making including treatment selection, sequencing strategies, and adherence to guidelines,” Saini adds. “This results in unwarranted variability in patient care. Also, reimbursement decisions become complicated since it is unclear whether a treatment is being used within its licensed indication.” By contrast, a clear and detailed understanding of the number, type and setting of prior lines of therapy impacts clinical outcomes and patient safety.
Beyond individual patient care, the standardisation proposed by EnLiST would have far-reaching implications for clinical research. “Without agreed definitions, it is effectively impossible to compare results across studies – a problem that is particularly pronounced in later-line treatment settings, where the complexity of prior therapy histories is greatest,” notes Koopman. “If one study defines a new LoT as any change in treatment, including drug discontinuation for toxicity or a treatment break, while another requires documented disease progression, the two studies are effectively enrolling different patient populations.”
Experts agree that ambiguity in LoT definitions can also result in incorrect interpretation of treatment efficacy and safety in clinical trials. “For example, a population on a second line in one study may be more heavily pretreated than in another, or outcomes may appear better or worse due to differences in prior therapy exposure, not the intervention itself. Also, lack of standardisation results in unclear inclusion/exclusion criteria, inconsistent patient selection across sites, and challenges in interpreting and reproducing trial results,” adds Saini.
By standardising definitions , EnLiST ensures that patient outcomes are comparable across institutions and settings. Given the wide variety of cytotoxic, hormonal, targeted, immune, and cellular treatments currently available, EnLiST was developed specifically around systemic therapies. “The question of extending EnLiST to loco-regional therapies is not closed,” concludes Koopman. “As the role of local ablative therapies in managing oligoprogressive disease continues to evolve, the framework may be reconsidered and updated in future versions to reflect emerging clinical practice.”