A study presented at ESMO Congress 2022 highlights the need to identify predictors of anticancer drug toxicity as more complex treatments become available
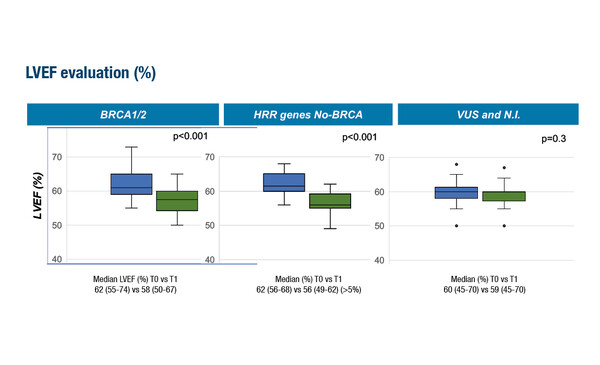
Data from a Mini Oral Session presentation at ESMO Congress 2022 suggest that pathogenic variants in homologous recombination repair (HRR) genes could increase the sensitivity of cardiac cells to anthracyclines in patients with breast cancer (Abstract 904MO). The multi-institutional study of 483 patients found that individuals carrying BRCA or other HRR deleterious genetic variants showed a statistically significant reduction in left ventricular ejection fraction (LVEF) >5% during monitoring and versus pre-treatment LVEF values (p<0.001).
Prof. Markus Joerger from Cantonal Hospital St. Gallen, Switzerland, found the findings of this study to be intriguing: “Anticancer drug-associated toxicity is often seen as unpredictable, and this is particularly true for less frequent types of adverse events. Pharmacogenomics have helped us to predict an increased risk of toxicity in some special cases including dihydropyrimidine dehydrogenase deficiency in the case of fluoropyrimidines, but advances in the field of predictive biomarkers for anticancer drug toxicity have been slow. The detection of HRR pathogenic germline variants as a risk factor for anthracycline-associated cardiotoxicity is important and opens the way for a better understanding of inter-individual variability in anticancer drug toxicity.”
Although the findings of this study presented at ESMO are hardly practice changing at this stage, they highlight the need to better understand and be able to predict toxicity which then impacts treatment safety. “This is a neglected field that deserves further attention,” adds Joerger, “much of the biomarker research in oncology is focused on predicting response to treatment, which is important, but we also need to be able to predict the associated toxicity.”
The expert explains that most patients who receive anthracyclines have some cardiac damage, although this can be minor in many cases. “The incidence of anthracycline-induced cardiac dysfunction (ACD) is approximately 6% for overt heart failure and up to 18% for subclinical cardiac dysfunction (Am J Cardiol. 2013;112:1980–1984). ACD has a poor prognosis, with published cardiovascular mortality rates of 9% at 5 years and 24% at 10 years (Eur J Heart Fail. 2018;20:898–906). Higher mortality rates have been reported at around 60% at 2 years in patients who develop anthracycline-induced cardiomyopathy (N Engl J Med. 2000;342:1077–1084).” ACD is usually classified as ‘early’ or ‘late’, with ‘early’ ACD developing within 12 months of treatment and ‘late’ ACD after several years (Eur Heart J. 2016;37:2768–2801). “However,” cautions Joerger, “it is clear that signs of cardiac damage, such as an increase in troponin, are already detectable during or early after anthracycline administration, indicating direct damage to the myocardium upon infusion. If serial echocardiographic assessment is performed, almost all asymptomatic heart dysfunctions are detected within the first year after anthracycline-containing chemotherapy.”
Decreasing treatment-associated toxicity can help to improve compliance, reduce hospitalisations and avoid potentially irreversible damage from anticancer agents. “In an era when treatments are becoming more complex and often include combinations of agents that can be associated with unexpected, rarer toxicities, improving the safety profile of regimens is becoming more and more important,” concludes Joerger, “also, with more treatments becoming available, it may be possible to consider alternative anticancer agents to replace anthracyclines in patients with HRR pathogenic variants or those otherwise deemed to be at higher risk of toxicity from a particular agent.”
Abstract presented:
Incorvaia L, et al. Anthracycline-related cardiotoxicity in breast cancer patients carrying mutational signature of homologous recombination deficiency (HRD). ESMO Congress 2022, Abstract 904MO
Mini Oral Session, 11.09.2022, h. 14:45 – 16:15, Dijon Auditorium