Results from two studies suggest that this approach is essential to refining liquid biopsy use in early cancer detection and to check tumour progression
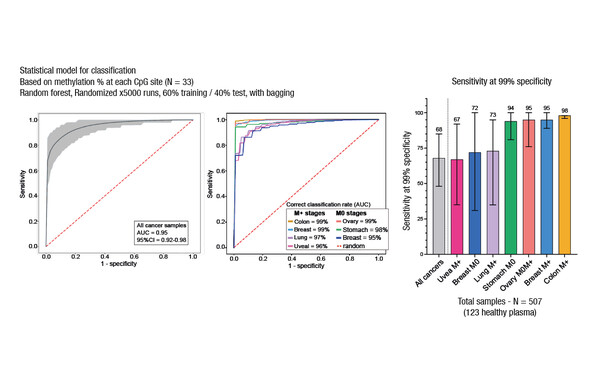
Presentations at the Molecular Analysis for Precision Oncology (MAP) Congress 2022 suggest that combining different liquid biopsy methodologies with other technologies may improve the limit of detection (LOD) and the reference range of liquid biopsies, with some implications for early cancer detection and monitoring of tumour progression. In a study from France, the use of a new highly sensitive methodology confirmed that hypomethylation of circulating retrotransposons will be valuable in the development of more efficient, non-invasive diagnostic tests across cancer types (Abstract 5MO). Hypomethylation of LINE-1 elements (L1PA), a shared feature of many cancers, were detectable in cell-free DNA (cfDNA) and discriminated well between plasma from 123 healthy individuals and 394 patients with a number of different cancers including metastatic colorectal, breast, lung and uveal cancers and non-metastatic tumours (area under the curve [AUC] 0.95). The results showed 67–98% sensitivity at 99% specificity across cancers. The robustness of LP1A hypomethylation as a marker was confirmed on an independent validation cohort of plasma from 30 healthy patients and 160 patients with cancers including metastatic breast, colorectal, gastric and lung cancers and non-metastatic ovarian cancers (AUC 0.99).
The group used a multiplex PCR-based targeted bisulfite method coupled to deep sequencing, along with computational tools to align sequencing data in a genome reference-free manner. Machine learning-based classifiers, integrating methylation patterns at single CpG sites and at the single molecule level, were used to discriminate between plasma from cancer patients and healthy individuals.
“Not only does this type of methylation analysis have the potential to be used as an agnostic test for the liquid biopsy-based detection of different types of solid tumours,” says Dr Umberto Malapelle from the Department of Public Health at the University of Naples Federico II, Italy, “but, crucially, the approach can be combined with other conventional liquid biopsy methods, like circulating tumour (ct) DNA mutational analysis, to improve the LOD of liquid biopsies. Improving the LOD is necessary to enable us to detect all clinically relevant variants in liquid biopsy, which is particularly important in the early disease setting.” Another important aspect of the study is the relatively simple nature of the methodology. “Because this method is already being used in other tissue-based settings and many laboratories already have the technology to apply to liquid biopsies, the approach has the potential to be adopted widely around the world,” says Malapelle. He also stresses that the fusion of information from the bioinformatic pipeline with data from the ‘wet’ biopsy, as demonstrated in the study, should be integral to the interpretation of liquid biopsies.
In a poster presentation, results from the TRACERx study in non-small-cell lung cancer (NSCLC) gave important insights into the use of longitudinal imaging and ctDNA tracking to map disease course and mechanisms of spread (Abstract 11P). Based on 436 imaging scans from 50 patients with post-surgical resection NSCLC relapse – including the lung (30%), lymph node (30%), bone (12%) and brain (5%) – ctDNA fraction did not always track with the total tumour volume. In two patients assessed, lesion-specific metastatic subclones detected at autopsy were absent in ctDNA, even though these subclones represented a large fraction of the total tumour volume on imaging, suggesting that many metastatic lesions do not shed significant amounts of ctDNA. Conversely, subclones from specific sites of disease were over-represented in ctDNA, suggesting a high rate of shedding.
The investigators tracked volumetric growth dynamics of individual metastatic lesions on serial images and contoured tumour dimensions using ITK-SNAP. ctDNA levels were detected with bespoke multiplex-PCR assay-panels based on tissue exome sequencing.
“The results from TRACERx highlight the importance of combining two different monitoring approaches, in this case, conventional imaging and liquid biopsy,” says Malapelle. “With this approach, we can gain a comprehensive view of the tumour progression. I hope to see more studies like this, which will eventually allow us to apply liquid biopsy to 3D tumour biology.” He continues, “The oncology community is aware that liquid biopsy can be used to predict relapse, resistance and tumour evolution, but we now need to have prospective trials to clinically validate the specific mutant allele fractions we are able to detect in liquid biopsies. The APPLE trial, which was presented at the ESMO Congress 2022 (Ann Oncol 2022:33[Suppl 7]:S808–S869), demonstrated the use of real-time PCR to monitor tumour progression in patients with EGFR-mutated NSCLC via the detection of ctDNA T790M. Future studies should be carried out using next-generation sequencing so that we can simultaneously monitor a number of different alterations, not just a single alteration.”
Abstracts presented:
Proudhon C, et al. Hypomethylation of circulating retrotransposons: towards a non-invasive pan-cancer diagnosis. MAP 2022, Abstract 5MO
Mini Oral Session 16.10.22, h. 15:40 – 16:40, Auditorium
Liu WK, et al. Mapping the pattern and pace of tumour dissemination using longitudinal imaging and ctDNA in the TRACERx lung study. MAP 2022, Abstract 11P
Poster Display Session 15.10.22, h. 12:15 – 13:00, Exhibition
Watch the session on demand on the Congress virtual platform