After proof of efficacy in patients with melanoma in a clinical trial, major efforts are now directed to make the use of TILs easier and safer in clinical practice.
New data on the long-term benefits of tumour-infiltrating lymphocytes (TILs), markers predictive of response and novel ways to avoid toxicity were presented at the ESMO Immuno-Oncology Congress 2023 (Geneva, 6–8 December), reinforcing the potential of TILs in advanced melanoma where improved outcomes versus ipilimumab have previously been shown (N Engl J Med. 2022;387:2113–2125).
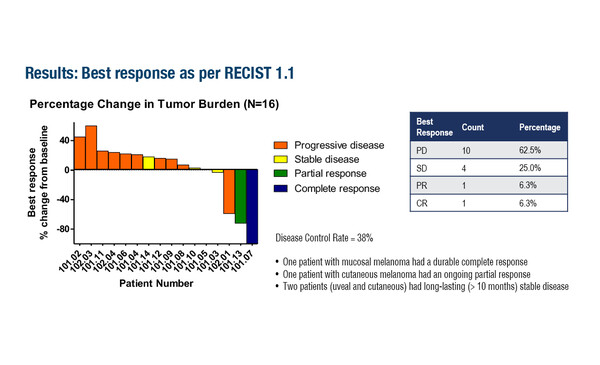
An earlier report from the phase II C-144-01 study demonstrated the benefits of lifileucel, a commercial, autologous TIL cell product, in patients with advanced melanoma previously treated with immune checkpoint inhibitors (ICI) and BRAF-/MEK-targeted agents (J Clin Oncol. 2021;39:2656–2666). As presented at the Congress, 4-year findings from the study show a sustained benefit with long-term survival in at least 20% of patients involved (Abstract 119O). In the analysis, treatment with lifileucel was associated with 1-, 2-, 3- and 4-year overall survival (OS) rates of 54.0%, 33.9%, 28.4% and 21.9% in 153 patients with advanced ICI-resistant melanoma. The objective response rate was 31.4% and the median duration of response was not reached. The highest 4-year OS rate was seen in patients with deepened responses (68.2%), but clinically meaningful rates were also achieved across all other patterns of response (early, late and not deepened). “It is remarkable that so many of these patients are still responding, given that this is a very difficult-to-treat population that has received all ICI options,” says Prof. John Haanen from the Netherlands Cancer Institute, Amsterdam, Netherlands. “The favourable effect seen in patients with deepened responses is very interesting, and this may act as a marker to describe a subset of patients deriving particular benefit from this approach.”
Another study presented in Geneva reported that complete metabolic response (CMR) on FDG-PET/CT scans may be a good predictor of long-term survival after TILs therapy in patients with advanced melanoma (Abstract 66MO). Among 44 patients, there was one complete response (CR), seven CMRs, 14 partial responses (PRs) and six partial metabolic responses (PMRs). Six of the seven patients achieving CMR were still alive and five of them were progression-free after more than 5 years. The median progression-free survival (PFS) in patients achieving PR was longer in those with CMR than in those with PMR (not reached versus 7.7 months; p=0.0003). “Predicting which patients will derive long-term efficacy from TILs is problematic,” says Haanen. “We know that patients who achieve a CR to treatment have the best outcomes. However, this may take time to develop and we also need to consider patients who achieve a PR – which of these will respond long term? This study suggests that a PET/CT scan could be used as a first line of evaluation in this setting.”
In a phase I trial, the oncolytic adenovirus TILT-123 – which is armed with tumour necrosis factor-alpha and interleukin-2 (IL-2) – was reported to be safe when used to enhance TILs cytotoxicity without the need for pre-conditioning chemotherapy or post-conditioning IL-2 in patients with ICI-resistant metastatic melanoma (Abstract 48O). TILT-123 was administered by intratumoural and intravenous injections and the most frequent adverse events related to it were fever (63%) and injection site pain (44%). The combination of TILT-123 and TILs did not increase the severity of adverse events. There was one durable CR, one ongoing PR and two cases of stable disease lasting >10 months. “The use of TILT-123 is an elegant proposition to deal with the toxicity – neutropenia, fever, thrombocytopenia – associated with traditional TILs therapy, most of which is the result of the conditioning regimen,” observes Haanen. “It is exciting to see, therefore, that this approach is feasible and that there are also signs of efficacy.”
Haanen concludes: “We know that TILs are here to stay in melanoma. The next challenge is how we can improve this treatment to make it easier and safer. Investigation of genetically modified TILs, such as those lacking PD-1 expression or those expressing cell surface IL-15, may provide useful ways of increasing efficacy and avoiding the toxicity associated with conditioning regimens. There is also a move to extend TILs to other cancer types that have poor sensitivity to ICIs and so are more difficult to treat, such as some cervical and lung cancers and rarer forms of melanoma. The results presented at the ESMO Congress 2023, showing a 50% response to lifileucel in mucosal melanoma, are very promising.”
Abstracts discussed:
Medina T, et al. Long-term efficacy and patterns of response of lifileucel tumor-infiltrating lymphocyte (TIL) cell therapy in patients with advanced melanoma: A 4-year analysis of the C-144-01 study. ESMO Immuno-Oncology Congress 2023, Abstract 119O
Proffered Paper Session 2, 07.12.2023, h. 14:15 – 15:45, Room B
Svane I-M, et al. Early complete metabolic response predicts long-term efficacy after adoptive cell therapy using tumor-infiltrating lymphocytes. ESMO Immuno-Oncology Congress 2023, Abstract 66MO
Mini Oral Session, 07.12.2023, h. 08:30 – 09:50, Room C
Monberg TJ, et al. Safety and efficacy of combined treatment with tumor infiltrating lymphocytes (TILs) and oncolytic adenovirus TILT-123 for patients with metastatic melanoma: Results from a phase I trial. ESMO Immuno-Oncology Congress 2023, Abstract 48O
Proffered Paper Session 2, 07.12.2023, h. 14:15 – 15:45, Room B