The role of neoadjuvant versus adjuvant immunotherapy is no simple comparison but instead involves a complex interplay of factors, based on tumour immune responsiveness, immunotherapy type, combination partners and trial design
Neoadjuvant therapy offers the opportunity to reduce the risk of disease relapse using short courses of treatment that are beneficial in terms of efficacy, patient quality of life and healthcare resource use. While fewer randomised studies have been conducted in the neoadjuvant compared with the adjuvant setting, current thinking is moving towards neoadjuvant treatment becoming the preferred approach. The counterintuitive strategy of retaining the tumour during treatment is based on sound rationale. Neoadjuvant use of checkpoint inhibitors activates both the priming phase of immunity within tumour tissue and the effector phase within the tumour microenvironment. This dual attack is elicited to a lesser extent if the macroscopic tumour has already been removed, as is the case for adjuvant therapy, and data show that neoadjuvant immunotherapy expands more T-cell clones than adjuvant treatment (Nat Med. 2018;24:1655–1661).
However, the clinical situation is a lot more complicated than just comparing ‘before’ with ‘after’ definitive treatment and applying it across the board. Firstly, immune responsiveness is well documented to vary between tumour types, even those arising at the same organ site. In a recent phase II trial, neoadjuvant treatment with the PD-1 inhibitor dostarlimab led to a 100% complete clinical response in 12 patients with microsatellite instability-high rectal cancer (N Engl J Med. 2022;386:2363–2376). In colorectal cancer, the pathological response rate to neoadjuvant treatment with the anti-CTLA-4 treatment ipilimumab plus the PD-1 inhibitor nivolumab was high in mismatch repair (MMR)-deficient disease but considerably lower in MMR-proficient disease (Nat Med. 2020;26:566–576). Also, an immunotherapeutic approach that is effective in one tumour type may not work as well in another, and for this we can contrast the efficacy of PD-1 blockade in melanoma (Lancet. 2021;398:1002–1014) with the failure of PD-L1 blockade in renal cell carcinoma (RCC) (JAMA Oncol. 2022;8:275–280).
Then there is the question of combination treatment. Combining immune therapies can provide additional benefit, which has been shown for many different cancers, but this is not guaranteed, as shown by the adjuvant CheckMate 915 trial, in which adding ipilimumab to adjuvant nivolumab failed to improve recurrence-free survival (RFS) in localised melanoma (Cancer Res. 2021;81:Suppl. 13:CT004). In contrast, adding ipilimumab to nivolumab did lead to improved RFS in an exploratory analysis of the IMMUNED study performed in patients with no evidence of disease after resection or radiotherapy of distant metastases (Lancet. 2020;395:1558–1568). However, overall survival data presented at ESMO Congress 2022 show a benefit of adjuvant ipilimumab–nivolumab over placebo but not over nivolumab alone (Abstract 784O). The authors reasonably suggest that the nivolumab alone vs placebo comparison may have been impacted by the high subsequent use of PD-L1 therapies in the placebo arm. Another presentation reports that Part A of the randomised, phase III CheckMate 914 trial did not meet the primary endpoint of disease-free survival for an adjuvant ipilimumab–nivolumab combination versus placebo post-nephrectomy in high-risk RCC (HR 0.92; 95% CI 0.71–1.20; p=0.5390) (LBA4).
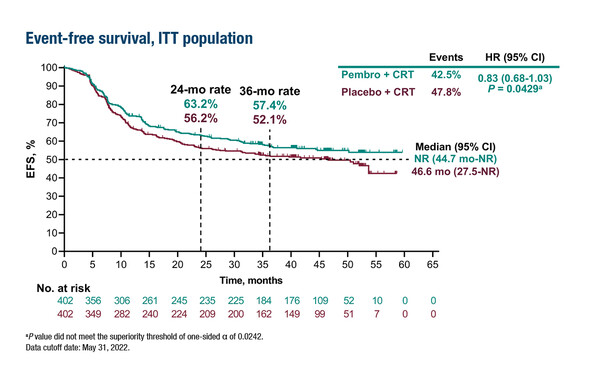
There is also the option to combine immunotherapy with other types of treatment – such as chemotherapy, targeted therapy and radiotherapy – many of which are current standards of care. This approach is being used in the phase III KEYNOTE-412 study, comparing neoadjuvant chemoradiotherapy with or without pembrolizumab; primary results from the study are being presented at the Congress (LBA5). However, combining immunotherapy with other immunotherapies or with different treatment approaches inevitably leads to greater toxicity and the need to balance anticipated benefit with potential harm including, but not limited to, the risk of delayed surgery.
The main obstacle hindering understanding of the relative benefits of neoadjuvant and adjuvant therapy lies in the design of clinical trials. Many trials comparing the two approaches are actually comparing neoadjuvant PLUS adjuvant therapy versus adjuvant therapy, making it difficult to unpick which element of treatment is giving the benefit. This is also the case with the SWOG S1801 study, presented in recent days and comparing neoadjuvant and adjuvant pembrolizumab in resected melanoma (LBA6). In order to determine the relative benefits of neoadjuvant and adjuvant approaches, the most important challenge now is to conduct well-designed, controlled trials that distinguish between the two components of treatment. The ongoing NADINA trial (NCT04949113), comparing neoadjuvant ipilimumab plus nivolumab with adjuvant nivolumab, should bring us closer to answering this question in melanoma.
Abstracts presented:
Patel S, et al. Neoadjuvant versus adjuvant pembrolizumab for resected stage III-IV melanoma (SWOG S1801). ESMO Congress 2022, LBA6
Presidential Symposium II, 11.09.2022, h. 16:30 – 18:15, Paris Auditorium
Machiels J-P, et al. Primary results of the phase III KEYNOTE-412 study: Pembrolizumab (pembro) with chemoradiation therapy (CRT) vs placebo plus CRT for locally advanced (LA) head and neck squamous cell carcinoma (HNSCC), LBA5
Presidential Symposium II, 11.09.2022, h. 16:30 – 18:15, Paris Auditorium
Motzer R, et al. Adjuvant nivolumab plus ipilimumab (NIVO+IPI) vs placebo (PBO) for localized renal cell carcinoma (RCC) at high risk of relapse after nephrectomy: Results from the randomized, phase III CheckMate 914 trial. ESMO Congress 2022, LBA4
Presidential Symposium II, 11.09.2022, h. 16:30 – 18:15, Paris Auditorium
Schadendorf D, et al. Adjuvant nivolumab (NIVO) alone or in combination with ipilimumab (IPI) versus placebo in stage IV melanoma with no evidence of disease (NED): Overall survival (OS) results of IMMUNED, a randomized, double-blind multi-center phase II DeCOG trial. ESMO Congress 2022, Abstract 784O
Proffered Paper Session – Melanoma and other skin tumours, 10.09.2022, h. 14:45 – 16:15, Fécamp Auditorium