Personalisation of treatment could be a step closer by integrating several cancer characteristics, but consolidation of data from these tools is needed before their use in clinical practice
At the ESMO Asia Congress 2023 (Singapore, 1–3 December), promising results were presented for novel tools that could help predict response and tailor treatment in patients with HER2-positive breast cancer.
Results from an effort to discover image-based biomarkers of response to the pan-AKT inhibitor, MK-2206, showed the potential of diffusions visible on dynamic contrast-enhanced (DCE)-MRI scans to predict pathological complete response (pCR) in patients with high-risk, early-stage HER2-positive and/or hormone receptor (HR)-negative breast cancer (Abstract 1MO). Previously, MK-2206 plus standard neoadjuvant chemotherapy showed promise for improving the pCR rate in patients enrolled in the I-SPY2 trial, although more than 40% of patients did not respond (J Clin Oncol. 2020;38:1059–1069). As presented in Singapore, researchers found one highly sensitive image biomarker – glcm_SumSquares (GLCM_SS) – that was predictive of response to MK-2206-based treatment in the entire biomarker-discovery cohort of 1,104 patients and in subgroups with HER2-negative, HR-negative and triple-negative disease, as well as those with a MammaPrint recurrence risk score of 2. GLCM_SS-positive disease was also associated with a ‘hot’ tumour microenvironment.
“The identification of imaging biomarkers could be advantageous, enabling the non-invasive selection of responders, helping to avoid unnecessary treatment and its associated toxicities, as well as potentially reducing costs to society,” says Dr Evandro de Azambuja from the Jules Bordet Institute, Brussels, Belgium. “While these results show promise for predicting response to MK-2206, they are hypothesis-generating findings, and further data are needed to build a dynamic prediction model by combining MRI characteristics with other markers.”
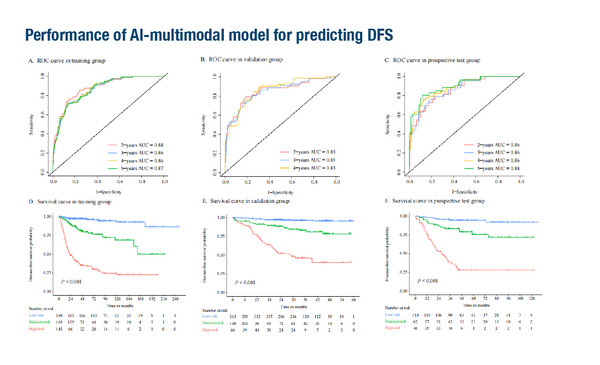
Other presentations revealed the results of studies integrating multiple biomarkers into models that could predict outcomes. The first of these used artificial intelligence (AI) techniques combining histopathology and clinical data to effectively predict pCR and disease-free survival (DFS) with neoadjuvant chemotherapy in 1,598 patients with breast cancer (Abstract 2MO). The researchers developed an AI-histopathology model, which then led to the development of a further AI multimodal model that fused histopathological and clinical information. The AI-histopathology model accurately predicted pCR in a training cohort of 756 patients, a validation cohort of 560 patients and a blinded prospective test cohort of 227 patients (area under the curve [AUC] values of 0.999, 0.995 and 0.981, respectively). The AI-multimodal model showed superior predictive ability to the AI-histopathology model, with respective AUC values of 0.999 and 0.994 in the validation and prospective test cohorts. Additionally, Kaplan–Meier analysis showed that patients predicted by the AI-multimodal model to achieve pCR had favourable DFS compared with patients who did not achieve pCR (p<0.05).
Another presentation highlighted an AI-assisted model based on an innovative multimodal approach integrating clinicopathological data with deep-learning MRI insights (3D-MMR model), (Abstract 3MO). The 3D-MMR model demonstrated a high degree of predictive accuracy for 4-year DFS, while using visualisation techniques, patients at high risk of metastases or recurrence were found to have differently located ‘hotspots’ compared with patients without such occurrences. Commenting on the AI models, de Azambuja says, “The key aspect of these two studies is that they looked at many features linked to the tumour microenvironment, including infiltration and functional status of T and B cells. The resulting models are dynamic and incorporate several biomarkers based on histopathology and clinical information to predict patient outcomes.”
Finally, a fourth presentation showed promising results with a prediction model for risk of lymphoedema in patients undergoing sentinel lymph node biopsy (Abstract 4MO). The study found that in a multivariate regression analysis, cytotoxic systemic chemotherapy (odds ratio [OR] 7.37), obesity (body mass index ≥30 kg/m2; OR 4.34), radiotherapy (OR 2.21) and the number of excised lymph nodes (OR 1.10) were independent risk factors significantly associated (p<0.05 for all factors) with the development of lymphoedema. “The risk of lymphoedema is lower with sentinel lymph node biopsy than axillary lymph node dissection, but being able to predict which patients are at high risk could aid in discussions with the patient about their treatment and side effects,” says de Azambuja. While informative, he points to clinical practice guidelines that provide specific recommendations on when sentinel lymph node biopsy or axillary lymph node dissection should be performed.
The future for personalisation of breast cancer treatment will stem from a combination of new tools like those described in Singapore, suggests de Azambuja. “Fusing imaging biomarkers, gene expression profiling, tumour microenvironment analysis and clinical characteristics will help us understand which patients will respond to neoadjuvant treatments. Each type of breast cancer will likely require its own prediction model and we need prospective data to back these models up,” he adds. Initiatives are underway in this respect, such as the European Organization for Research and Treatment of Cancer (EORTC) SPECTA platform, with recent preliminary results showing the possibility of integrating multiple data layers using different deep learning approaches to predict pCR from baseline samples from triple-negative breast cancer and HER2-positive neoadjuvant patient cohorts in the prospective IMMUcan study (Ann Oncol. 2023;34(Suppl. 2):S1167).
Abstracts discussed:
Zhang J, et al. Image biomarker discovery from DCE-MRI for identifying responders of MK-2206 on early-stage breast cancer patients: A secondary radio-genomics analysis of I-SPY2 trial. ESMO Asia Congress, Abstract 1MO
Mini Oral Session: Breast Cancer, 03.12.2023, h. 10:45 – 12:15 SGT, Hall 401
Yu Y, et al. Multimodal data fusion enhanced precision neoadjuvant chemotherapy in breast cancer with a multi-task transformer-CNN-mixed learning. ESMO Asia Congress, Abstract 2MO
Mini Oral Session: Breast Cancer, 03.12.2023, h. 10:45 – 12:15 SGT, Hall 401
Ren W, et al. Multimodal data fusion for improved risk stratification of breast cancer with multi-task 3D deep learning model: A multicenter study. ESMO Asia Congress, Abstract 3MO
Mini Oral Session: Breast Cancer, 03.12.2023, h. 10:45 – 12:15 SGT, Hall 401
Byeon JY, et al. A prediction model for developing lymphedema in breast cancer patients receiving sentinel node biopsy. ESMO Asia Congress, Abstract 4MO
Mini Oral Session: Breast Cancer, 03.12.2023, h. 10:45 – 12:15 SGT, Hall 401