In two studies, the use of ESCAT matching and ESMO recommendations for germline assessment lead to effective targeted treatment
As presented at the ESMO Targeted Anticancer Therapies (TAT) Congress 2026 (Paris, 16–18 March), ESCAT tier I and tier II matched targeted therapies demonstrated meaningful antitumour activity in the 360 RESISTANCE study, which is evaluating outcomes and mechanisms of resistance to targeted therapies in early-phase clinical trials (Abstract 137MO).
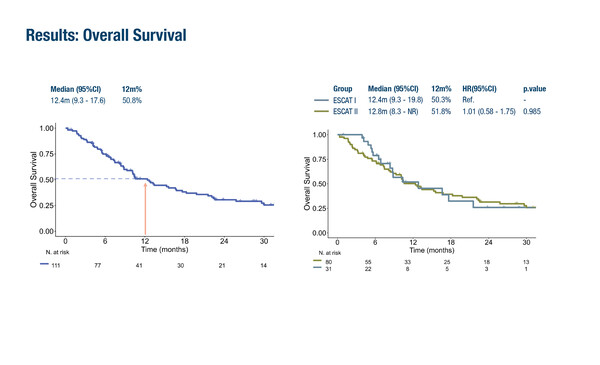
Findings from the prospective observational trial highlight the value of molecular pre-screening programmes. As reported by researchers, the overall response rate (ORR) was 39% in 111 patients categorised as ESCAT I or ESCAT II who went on to receive matched targeted therapy in early-phase trials at the Vall d'Hebron Institute of Oncology (VHIO) between 2015 and 2025. The ORR was similar for ESCAT I (41%) and ESCAT II (32.3%; p=0.512). Median progression-free survival was 5.4 months, median overall survival (OS) was 12.4 months and the 12-month OS rate was 50.8%.
The most prevalent tumour types were colorectal (27%), lung (15%) and biliary tract (13%) cancers. Targets were identified on tissues (88%) or plasma (12%) and genome matched with DNA via next-generation sequencing (NGS) (76%) or RNA/immunohistochemistry (24%). The most frequent alterations were KRAS (24%), BRAF (16%), HER2 (14%), RET (12%) and NTRK (10%). Grade 3–4 treatment-related adverse events (TRAEs) were reported in 31% of all patients, with no significant difference between the rates observed for ESCAT I (31%) and ESCAT II (29%).
“This study confirms that when patients with actionable molecular alterations high in the ESCAT rankings are treated with appropriate targeted therapies, the outcomes are good, underlining the validity of the scale and the recommendation to screen all patients for these relevant alterations,” comments Dr Elena Castro from Hospital Universitario 12 de Octubre, Madrid, Spain. “Also, ESCAT I and II categories are defined separately based on the available evidence regarding clinical benefit, but the study suggests little difference between them in outcomes with the targeted therapies used.” She observes, however, that: “The benefits of using ESCAT can only be fully realised if there is sufficient access to suitable therapies and this is a problem for many centres, for example due to a lack of regulatory approval or to reimbursement issues.” Ensuring that ESCAT evolves dynamically as new targets and treatments emerge was also noted as key to its continued success.
In another, real-world, retrospective study from VHIO’s molecular pre-screening programme, only 39.1% of patients who fulfilled the ESMO criteria for germline assessment were evaluated by genetics experts (Abstract 152P). Of the 31.0% of patients who progressed to germline testing, 63.8% were confirmed to carry germline pathogenic variants. Altogether, 32.8% of the patients with BRCA1 or BRCA2 alterations underwent germline testing, with germline origin being confirmed in 93.8% and 97.9%, respectively, of variants. The analysis included 562 patients undergoing tumour-only NGS between 2020 and 2025.
“This study provides disappointing evidence that despite ESMO recommendations on germline-focused analysis (Ann Oncol. 2019;30:1221–1231) many eligible patients – 60% in the case of this study – are missing out on expert genetic referrals, including those with highly actionable alterations,” notes Castro. “The detriment to these patients and their families is highlighted by the study’s high rate of germline pathogenic variant confirmation among referrals.” The reason patients are not referred is unclear, but the authors of the study suggest that advanced stage at tumour-only testing, resulting in an insufficient time to act, may be a contributing factor.
Castro thinks that a lack of awareness of the recommendations may also be an issue and concludes that “greater efforts are needed to ensure that the benefits of germline testing are made available to all eligible patients.”
Programme details
Bruzzone F, et al. Outcomes of patients (pts) treated with ESCAT I–II targeted therapies (TT) in early phase clinical trials (EPCTs). ESMO Targeted Anticancer Therapies 2026 - Abstract 137MO
Aguilar Izquierdo S, et al. Adoption of ESMO guidelines for germline testing after tumour-only sequencing in patients (pts) with advanced cancers. ESMO Targeted Anticancer Therapies 2026 - Abstract 152P