Early activity and manageable safety were demonstrated in two phase I trials with innovative agents
CD47 functions as a ’don’t eat me‘ signal, preventing natural phagocytosis of healthy cells such as erythrocytes and platelets by interacting with SIRPα expressed on macrophages, dendritic cells and neutrophils (Int J Mol Sci. 2025;26:5394). It is frequently overexpressed in malignancies, functioning as an innate immune checkpoint and enabling tumour cells to promote immune evasion. Targeting CD47-SIRPα alone or simultaneously with other mechanisms has the potential to promote antitumour efficacy as suggested by two phase I studies presented at the ESMO Targeted Anticancer Therapies (TAT) Congress 2026 (Paris, 16–18 March).
The feasibility of triple-checkpoint targeting was demonstrated with HCB301, a novel SIRPα-PD-L1-TGF-β fusion protein (Abstract 60O). “Preclinical evidence indicates that HCB301 engages with SIRPα, PD-L1 and TGF-β, suggesting that innate, adaptive and stromal checkpoint targeting are feasible with one molecule,” notes Dr Alberto Hernando-Calvo from the Vall d’Hebron Institute of Oncology (VHIO), Barcelona, Spain. Haematological toxicities were predictable and manageable with the doses of HCB301 tested (0.3, 0.6 and 1.2 mg/kg) in 19 heavily pre-treated patients with a range of solid tumour types. Treatment-related adverse events (TRAEs) were mostly grade 1–2 (86.5%). Two dose-limiting toxicities (DLTs) of grade 4 thrombocytopenia were observed at the highest dose studied. Stable disease was observed in 5 out of 14 evaluable patients who received HCB301 0.6–1.2 mg/kg with different tumour types. “For now, the TRAEs appear manageable with this triple hit, however we will need to see more granularity in the safety and efficacy data as doses are escalated,” he cautions. Dose escalation now continues to determine the recommended phase II dose.
On-target, off-tumour toxicity has previously been a key limitation to the clinical advancement of CD47 antibodies (PLoS One. 2019;14:e0218897). A second study presented in Paris found promising preliminary findings with HCB101, a SIRPα–IgG4 Fc fusion protein designed to have reduced erythrocyte binding but preserved CD47 targeting on tumour cells (Abstract 61O). “There appears to be a reduced rate of the cytopenias that have previously limited attempts to target SIRPα, although again, more details on the safety profile are needed. There are also early signs of anti-tumour activity with HCB101 as monotherapy, supporting future combination strategies,” notes Hernando-Calvo.
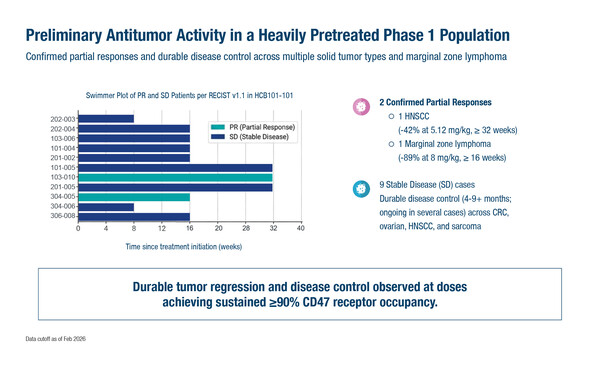
Across the HCB101 dose range of 0.08–36 mg/kg, the maximum tolerated dose was not reached. No grade ≥3 anaemia was observed and there was no signal for cumulative anaemia across the dose levels. Two DLTs were observed: grade 3 and grade 4 thrombocytopenia but with no bleeding-associated events. Two patients had confirmed partial responses – one with head and neck cancer (HCB101 5.12 mg/kg) and one with marginal zone lymphoma (HCB101 8 mg/kg). Nine patients with different tumour types had stable disease. Sustained ≥90% CD47 receptor occupancy was observed at doses of 8 mg/kg or above.
“Preliminary findings for both HCB301 and HCB101 show promise, but it will be important to analyse efficacy data by previous exposure and response to immunotherapy to establish their potential place in the treatment landscape. Positive correlative studies will also aid in the identification of predictive biomarkers,” concludes Hernando-Calvo.
HCB101 is currently being tested in combination with other treatments including chemotherapy, anti-PD-1 antibodies, anti-VEGF antibodies, HER2-targeted agents and EGFR-based regimens (NCT06771622; J Clin Oncol. 2026;44(Suppl.):372). Other anti-SIRPα antibodies in early phase development include evorpacept (Blood. 2025;146 (Suppl.1):3571) and DS-1103a (NCT05765851).
At a glance:
Zhu J, et al. First-in-Human Phase 1 Evaluation of HCB301, a Tri-specific SIRPα-PD-1-TGFβ Fusion Protein: Safety, Pharmacokinetics, and Multisystem Immune Activation. ESMO Targeted Anticancer Therapies Congress 2026 - Abstract 60O
- N=19 (14 efficacy-evaluable pts)
- DLTs: 2 grade 4 thrombocytopenias
- TRAEs: mostly grade 1−2
- Stable disease: 36%
- Progressive disease: 64%
Ning F, et al. First-in-Human Phase 1 Evaluation of HCB101, a SIRPα–Fc Innate Checkpoint Fusion Protein: Safety, Pharmacokinetics, and Receptor Occupancy. ESMO Targeted Anticancer Therapies Congress 2026 - Abstract 61O
- N=67 (59 DLT-evaluable pts)
- DLTs: 1 grade 3 thrombocytopenia and 1 grade 4 thrombocytopenia
- TRAEs: 84.0% were grade 1−2
- Partial response: 2 pts
- Stable disease: 9 pts