Preliminary data on the tetravalent DR5 agonist indicate strong and durable responses with a manageable safety profile
Results from a phase I trial with ozekibart added to standard chemotherapy indicate encouraging efficacy and safety in adolescents and adults with Ewing sarcoma, according to a presentation at the ESMO Sarcoma and Rare Cancers Congress 2026 (Lugano, 12–14 March) (Abstract 84MO). Ozekibart is a next-generation tetravalent agonistic antibody that targets death receptor 5 (DR5), expressed on the surface of bone and soft tissue sarcoma cells, enabling cancer-biased cell death (Clin Cancer Res. 2023;29:2988−3003; Front Mol Biosci. 2024:11:1384795). Ozekibart has demonstrated efficacy in preclinical models (Clin Cancer Res. 2023;29:2988−3003; Neuro-Oncology 2025;27(Suppl. 5)v362−v363) and notably, improved progression-free survival versus placebo when given as monotherapy in the ChonDRAgon phase II trial in patients with chondrosarcoma (ESMO Rare Cancers 2025;4:100116).
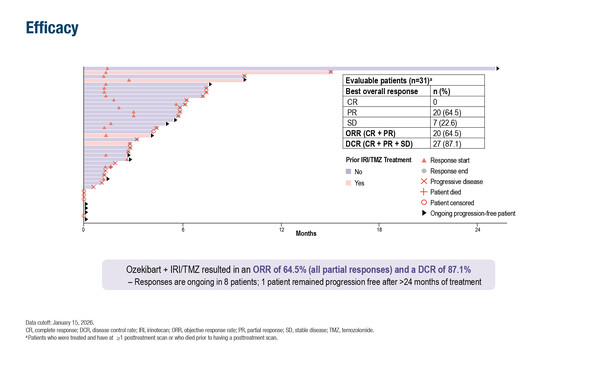
As presented at the Congress, treatment with ozekibart added to irinotecan and temozolomide resulted in an objective response rate (ORR) of 64.5% (all partial responses) in 31 evaluable patients with relapsed/refractory classical Ewing sarcoma, including those who had disease progression on prior irinotecan–temozolomide regimens. The disease control rate was 87.1% and one patient remained progression free after more than 2 years of treatment.
Commenting on the data, Prof. Herbert Loong from the Chinese University of Hong Kong, Hong Kong SAR, China, notes: “With few treatment options, prognosis is poor for patients with recurrent Ewing sarcoma and the observed response rate seen with the addition of ozekibart is very promising, particularly the signal for response observed in patients with prior irinotecan and temozolomide exposure.”
Hepatotoxicity has been a concern with previous attempts to target DR5 (Cancer Chemother Pharmacol. 2015;75:887–895). However, no grade ≥3 hepatotoxicity was observed with ozekibart in the presented study, in line with the low incidence of treatment-related hepatic events in the phase II trial in chondrosarcoma (ESMO Rare Cancers 2025;4:100116). “We have no head-to-head data with combination therapy but the safety profile with the triple combination raises no apparent concerns, although it would be of interest to understand the reasons for discontinuation of ozekibart,” says Loong. Overall, adverse events (AEs) with the investigated regimen were consistent with the known safety profile of irinotecan and temozolomide, most commonly, diarrhoea (grade ≥3: 35.9%), anaemia (grade ≥3: 20.5%), vomiting (grade 3: 10.3%) and nausea (grade ≥3: 7.7%). Four patients (10.3%) had AEs resulting in discontinuation of ozekibart.
Further evaluation of ozekibart in recurrent Ewing sarcoma and in other difficult-to-treat cancers where DR5 is overexpressed are now needed to confirm these promising findings. Enrolment in the phase I trial continues (NCT03715933).
Programme details
Chugh R, et al. Phase I study of the tetravalent death receptor 5 (DR5) agonist ozekibart (INBRX-109) combined with irinotecan (I) and temozolomide (T) in adolescents and adults with Ewing sarcoma. ESMO Sarcoma and Rare Cancers Congress 2026 - Abstract 84MO