Adding first-line checkpoint inhibition to standard treatment shows promise to improve response in women
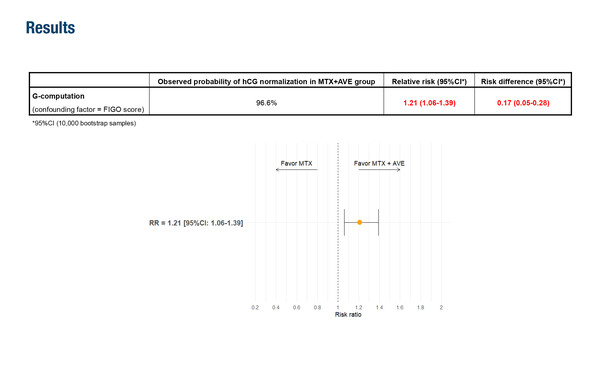
Results from a new analysis presented at the ESMO Sarcoma and Rare Cancers Congress 2026 (Lugano, 12–14 March) highlight a 21% improvement in complete response with the addition of the anti-PD-L1 antibody avelumab to standard treatment (8-day methotrexate) compared with standard treatment alone in patients with low-risk gestational trophoblastic neoplasia (GTN) (Abstract 31MO). The study compared data on serum human chorionic gonadotropin (hCG) normalisation allowing treatment discontinuation (complete response) from 29 patients who received avelumab plus 8-day methotrexate with data from an external control group of 247 patients who received 8-day methotrexate alone, collected over 5 years. In patients treated with additional immunotherapy, the observed probability of hCG normalisation was 96.6%, with a risk ratio of 1.21 (95% confidence interval [CI] 1.06–1.39) versus methotrexate alone based on a target trial emulation framework.
About 95% of patients with a hydatidiform mole who develop GTN are low risk and they usually respond well to standard therapy (Ann Oncol. 2013;24(Suppl. 6):vi39−50). Recently, there has been considerable excitement about the role of checkpoint inhibition to improve response further. The benefits of avelumab were initially observed in the second-line setting (J Clin Oncol. 2020;38:3129–3137) before positive findings were reported with first-line avelumab plus 8-day methotrexate in the single-arm phase I/II TROPHAMET trial (Ann Oncol. 2024;35(Suppl. 2):S545).
“This encouraging new study extends insights, providing a positive indirect comparison with methotrexate alone, thereby overcoming the challenge of acquiring comparative data in rare cancers,” comments Prof. Linda Mileshkin from the Peter MacCallum Cancer Centre, Melbourne, Australia. “However, we need the results to be confirmed by other large GTN databases and, ideally, by a randomised phase III trial. As clinicians, we know what to expect with the relatively inexpensive and effective 8-day methotrexate regimen. It will be important to demonstrate that improved cure rates with avelumab are balanced against the risk of long-term toxicity, including making sure that there is no adverse impact on fertility given some preclinical data about this (Nat Cancer. 2022;3:1–13).”
The question of which regimen to use following primary treatment resistance was explored in a UK registry-based study presented at the Congress. Multiagent chemotherapy (etoposide, methotrexate, actinomycin D, cyclophosphamide and vincristine [EMA/CO]) was associated with higher achievement of normal serum hCG for 1 year (sustained remission) than actinomycin D alone in 30 patients who developed primary methotrexate resistance (Abstract 42P). Sustained remission was observed in 91.3% (21/23) of patients receiving EMA/CO and 28.6% (2/7) receiving actinomycin D (odds ratio 26.25; 95% CI 3.50–234.36; p=0.003). Furthermore, significantly lower resistance rates were seen with EMA/CO versus actinomycin D (4.3% versus 42.9%, respectively; p=0.03).
“Although the numbers are small, there is a clear signal here for better response with the early second-line use of EMA/CO, an approach that is currently often delayed in the clinic,” notes Mileshkin. EMA/CO may be associated with more toxicity than single-agent chemotherapy so a frank discussion with the patient is needed. “Again, confirmation of the study’s findings using data from another of the international GTN registries would be ideal, as conducting a traditional clinical trial is challenging due to difficulties recruiting sufficient numbers of patients with primary methotrexate resistance,” comments Mileshkin. She concludes: “The next step in improving the treatment landscape for low-risk GTN is further biomarker analyses to predict the best first- and second-line regimen for each patient – balancing what is the most active regimen in any scenario against using treatments that have little, if any, potential for long-term side effects for this young patient population.”
Programme details:
Bolze P-A, et al. Comparing efficacy of avelumab + methotrexate vs. methotrexate alone in low-risk gestational trophoblastic neoplasia: a national registry-based external control comparison. ESMO Sarcoma and Rare Cancers Congress 2026 - Abstract 31MO
Charfare F, et al. Low risk gestational trophoblastic neoplasia with primary resistance to methotrexate should be treated with multi-agent chemotherapy rather than single agent actinomycin D. ESMO Sarcoma and Rare Cancers Congress 2026 - Abstract 42P